Chapter: Modern Pharmacology with Clinical Applications: Androgens, Antiandrogens, and Anabolic Steroids
Steroidogenesis
STEROIDOGENESIS
The main steroidogenic
components of the testis are the interstitial cells of Leydig found between the
seminifer-ous tubules. The principal secretory product of Leydig cells,
testosterone, is not stored to any significant degree within these cells.
Biochemical studies of Leydig cell steroidogenic function have shown that testosterone synthesis begins with acetate derived either from glucose or products
of lipid metabolism. Acetate is converted to cholesterol through numerous reactions in or on the smooth
endoplasmic reticulum. Cholesterol, once formed, is stored in lipid droplets in
an esterified form. The cholesterol required for steroidogenesis is
trans-ferred into the mitochondria, where the side chain is cleaved by enzymes
on the inner membranes to form pregnenolone. This reaction is the rate-limiting step in testosterone biosynthesis and is the step stimulated by luteinizing
hormone (LH). Pregnenolone is then re-turned to the cytoplasm, where it
serves as the principal precursor of testosterone.
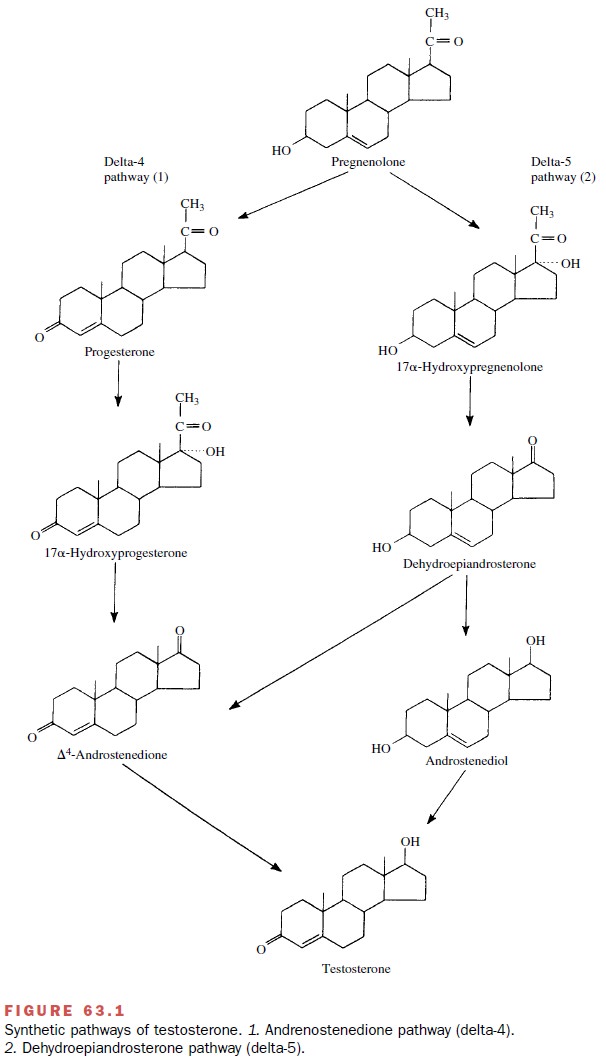
Testosterone synthesis from
pregnenolone can oc-cur along two distinct metabolic pathways (Fig. 63.1). The
names given to these two routes of metabolism re-fer to the position in the
steroid molecule where an un-saturated bond is maintained. Thus, in the delta-4
path-way an unsaturated position is between C4 and C5 of ring A, whereas in the
delta-5 pathway, the unsaturated position is between C5 and C6 of ring B. In the human testis, the delta-5 pathway is the predominant (but not ex-clusive) one
used for the biosynthesis of testosterone.
Sertoli cells, in the
seminiferous tubule wall, are known to be important in spermatogenesis, in part
through their synthesis of an androgen-binding
protein (ABP). ABP, when secreted
into the lumen of the sem-iniferous tubules, selectively binds testosterone of
Leydig cell origin and serves as a hormone reservoir and transport protein for
the androgen.
Related Topics