Chapter: Medical Immunology: Antigen-Antibody Reactions
Specific Types of Antigen-Antibody Reactions
SPECIFIC TYPES OF ANTIGEN-ANTIBODY REACTIONS
Antigen-antibody reactions may be revealed by a variety of physical expressions, depend-ing on the nature of the antigen and on the conditions surrounding the reaction.
A. Precipitation
When antigen and antibody are mixed in a test tube in their soluble forms, one of two things may happen: both components will remain soluble or variable amounts of Ag Ab precipi-tate will be formed. If progressively increasing amounts of antigen are mixed with a fixed amount of antibody, a precipitin curve can be constructed (Fig. 8.5). There are three areas to consider in a precipitin curve:
· Antibody excess—free antibody remains in solution after centrifugation of Ag Ab complexes.
· Equivalence—no free antigen or antibody remains in solution. The amount of pre-cipitated Ag-Ab complexes reaches its peak at this point.
· Antigen excess—free antigen is detected in the supernatant after centrifugation of Ag-Ab complexes.
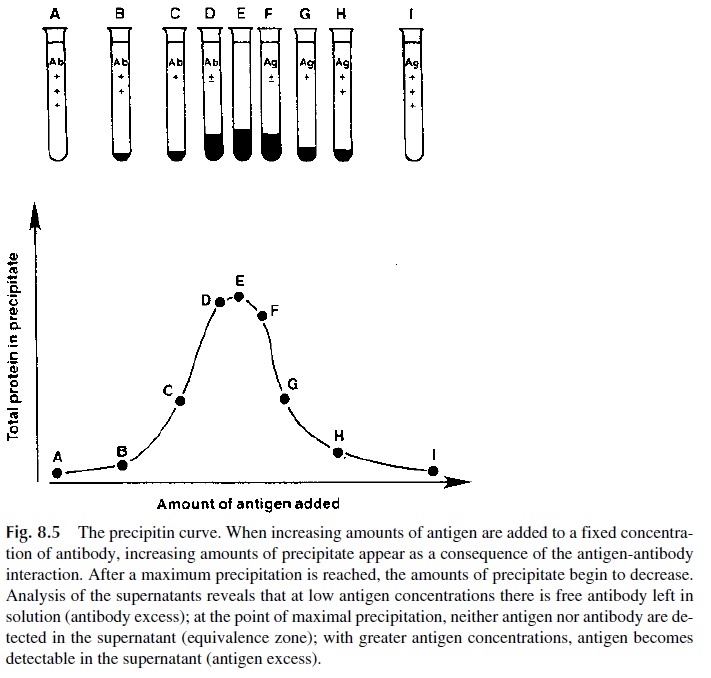
The “lattice theory” was created to explain why different amounts of precipitation are observed at different antigen-antibody ratios. At great antibody excess, each antigen will tend to have its binding sites saturated, with antibody molecules bound to all its exposed determinants. Extensive cross-linking of antigen and antibody is not possible. On the other hand, if one can determine the number of antibody molecules bound to one single antigen molecule, a rough indication of the valency (i.e., number of epitopes) of the antigen will be obtained. At great antigen excess, single antigen molecules will saturate the binding sites of the antibody molecule and not much cross-linking will take place either. If the antigen is very small and has no repeating epitopes, calculation of the number of antigen molecules bound by each antibody molecule will indicate the antibody valency, as discussed earlier. When the concentrations of antigen and antibody reach the equivalence point, maximum cross-linking between Ag and Ab will take place, resulting in formation of a large precipitate that contains all antigen and antibody present in the mixture (Fig. 8.6).

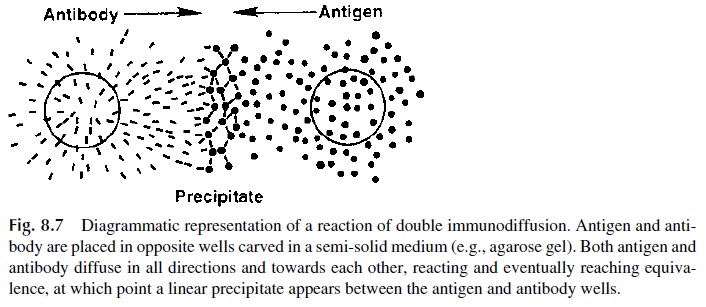
Semi-solid supports, such as agar gel, in which a carbohydrate matrix functions as a container for buffer that fills the interstitial spaces left by the matrix, have been widely used for the study of antigen-antibody reactions.
Antigen and antibody are placed in wells carved in the semi-solid agar and allowed to passively diffuse. The diffusion of antigen and anti-body is unrestricted, and in the area that separates antigen from antibody the two reactants will mix in a gradient of concentrations. When the optimal proportions for Ag-Ab binding are reached, a precipitate will be formed, appearing as a sharp, linear opacity (Fig. 8.7).
B. Agglutination
When bacteria, cells, or large particles in suspension are mixed with antibodies directed to their surface determinants, one will observe the formation of large clumps; this is known as an agglutination reaction.
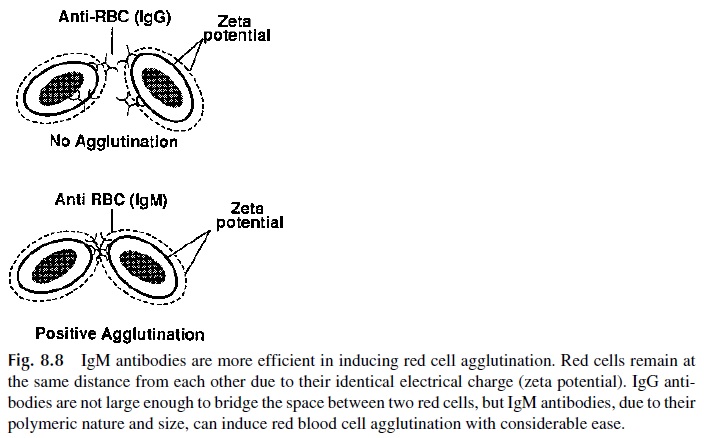
Agglutination reactions result from the cross-linking of cells and insoluble particles by specific antibodies. Due to the relatively short distance between the two Fab fragments, 7S antibodies (such as IgG) are usually unable to bridge the gap between two cells, each of them surrounded by an electronic “cloud” of identical charge that will tend to keep them apart. IgM antibodies, on the other hand, are considerably more efficient in inducing cellu-lar agglutination (Fig. 8.8).
The visualization of agglutination reactions differs according to the technique used for their study. In slide tests, the nonagglutinated cell or particulate antigen appears as a ho-mogeneous suspension, while the agglutinated antigen will appear irregularly clumped. If antibodies and cells are mixed in a test tube, the cross-linking of cells and antibodies will result in the diffuse deposition of cell clumps in the bottom and walls of the test tube, while the nonagglutinated red cells will precipitate in a very regular fashion, forming a compact red button on the bottom of the tube.
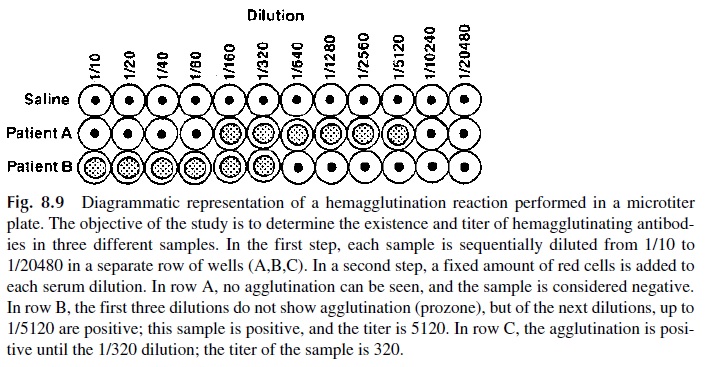
Agglutination reactions follow the same basic rules of the precipitation reaction. When cells and antibody are mixed at very high antibody concentrations (low dilutions of antisera), antibody excess may result, no significant cross-linking of the cells is seen, and, therefore, the agglutination reaction may appear to be negative. Dilutions in which anti-body excess prevents agglutination constitute the prozone.With increasing antibody dilu-tions, more favorable ratios for cross-linking are reached, and very fine clumps cover the walls of the test tube or microtitration wells. When equivalence is approached, larger clumps of cells can be distinguished. At still higher dilutions, when the concentration of an-tibody is very low, the zone of antigen excess is reached and agglutination is no longer seen (Fig. 8.9).
Related Topics