Chapter: Surgical Pathology Dissection : Lymph Nodes
Sentinel Lymph Node Biopsy
Sentinel Lymph Node Biopsy
As noted
above, the standard pathologic practice for the evaluation of nonsentinel lymph
nodes is to examine microscopically one section from each lymph node using the
simple H&E technique. Although this approach may be practical for
evaluating large numbers of lymph nodes, most pathologists would concede that
such limited analysis consistently underestimates the true in-cidence of occult
nodal metastases. Recent im-provements in our clinical ability to identify the
lymph nodes most likely to harbor metastases have facilitated the accurate
staging of cancers. The sentinel lymph node strategy is particularly appealing
because the surgical removal of just one or several selected lymph nodes
permits a more comprehensive pathologic search for small and localized
metastatic deposits.
Methods
for detecting tumor cells in sentinel lymph nodes have become increasingly
sophis-ticated and sensitive, ranging from routine histologic examination of
serial sections to re-verse transcriptase-polymerase chain reaction-based
methods for detecting a single tumor cell among a sea of lymphocytes. Outside
of routine H&E staining, however, most detection methods are
investigational, and currently there is no agreement as to an optimal detection
protocol. Given the diversity of the processing and exami-nation of sentinel
lymph node biopsies among laboratories, you should be familiar with the
protocol details specific to your own institution. At the same time, there are
generic guidelines that are widely applicable across institutions and assorted
tumor types.
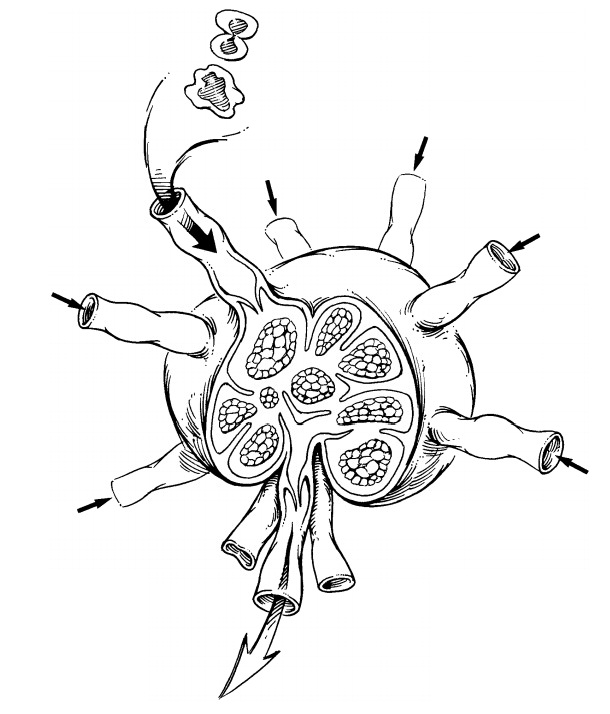
The sentinel
lymph node biopsy specimen should be carefully examined to determine the number
of lymph nodes. The size of each should be recorded. Each lymph node should be
pro-cessed separately. Each node is serially sectioned along the longitudinal
or transverse plane into 3- to 4-mm slices. Small lymph nodes that can-not be
easily sectioned should be submitted in toto. Examine the cut surface of each
slice for the presence of grossly visible tumor nodules.
Your
gross assessment of the lymph node dic-tates the degree of sectioning by the
histopathol-ogy laboratory. If tumor is visualized grossly, routine H&E
staining of a single level is sufficient to document the presence of tumor and
its possi-ble extension beyond the lymph node capsule. If tumor is not grossly
visible, the lymph node slices should be sectioned at multiple levels. There is
currently no standard to guide the ex-tent of tissue sectioning. At a minimum,
one section from each of three levels of the tissue block should be obtained
for routine H&E staining.Regardless of whether immunohisto-chemistry is
part of a specific protocol, the his-topathology laboratory should place
intervening unstained sections on sialinated slides in an effort to minimize
loss of potentially diagnostic material and provide a source of unstained
sections should the need for immunohisto-chemistry arise.
Handling Radioactive Specimens Obtained by Sentinel Lymphadenectomy
The
process of clinical lymphatic mapping and the identification of sentinel lymph
nodes relies on nodal uptake of radioactive tracers. Fortunately for pathology
personnel, the amount of radiation associated with sentinel lymphadenectomies
is low. Even with frequent handling of these speci-mens, radiation exposure
usually does not ap-proach statutory exposure limits. Given the exceedingly low
radiation exposure, most au-thorities now agree that quarantining these
specimens does not enhance the safety of pathol-ogy personnel and only serves
to delay the final diagnosis. Accordingly, sentinel lymph node biopsies should
be processed immediately on re-ceipt from the operating room using customary
universal precautions. Nonetheless, if you have a question about a particular
specimen, you should call your institution’s radiation safety officer.
Sentinel Lymph Node Biopsy for Evaluating Metastatic Disease
1. Record the number of lymph nodes and their dimensions.
2. Serial
section each lymph node along its longitu-dinal or transverse plane into 3- to
4-mm slices.
3. If a metastatic implant is grossly visible, have the histopathology laboratory cut and stain one representative section to document the presence of tumor.
4. If a
metastatic implant is not grossly visible, have the histopathology laboratory
cut multi-ple sections from at least three levels. At least one section from
each level should be stained with H&E. Additional unstained sections should
be stored on treated slides for future immunohistochemical studies as needed.
Important Issues to Address in Your Surgical Pathology Report
·
What procedure was performed?
·
How many lymph nodes from each anatomic level
harbor metastatic tumor, and how many lymph nodes from each level were
microscopi-cally examined?
·
What is the size of the largest metastatic
implant?
·
Does the metastasis extend beyond the nodal
capsule into the surrounding perinodal fat? (This is particularly important to
note for meta-static squamous cell carcinomas of the head and neck and
metastatic carcinomas of the breast.)
·
For sentinel lymph node biopsies, was the
metastasis detected by routine histopathol-ogy, immunohistochemistry,
molecular-genetic analysis, or some combination of these tech-niques?
Related Topics