Chapter: Modern Pharmacology with Clinical Applications: Mechanisms of Drug Action
Second-Messenger Systems
SECOND-MESSENGER
SYSTEMS
Many receptors are capable of
initiating a chain of events involving second messengers. Key factors in many
of these second-messenger systems are proteins termed G proteins, short for guanine nucleotide– binding proteins. G
proteins have the capacity to bind guanosine triphosphate (GTP) and hydrolyze
it to guanosine diphosphate (GDP).
G proteins couple the
activation of several different receptors to the next step in a chain of
events. In a num-ber of instances, the next step involves the enzyme adenylyl
cyclase. Many neurotransmitters, hormones, and drugs can either stimulate or
inhibit adenylyl cy-clase through their interaction with different receptors;
these receptors are coupled to adenylate cyclase through either a stimulatory
(GS) or an inhibitory (G1) G protein. During the coupling
process, the binding and subsequent hydrolysis of GTP to GDP provides the
en-ergy needed to terminate the coupling process.
The activation of adenylyl
cyclase enables it to cat-alyze the conversion of adenosine triphosphate (ATP)
to 3 5 -cyclic adenosine monophosphate (cAMP), which in turn can activate a
number of enzymes known as ki-nases. Each
kinase phosphorylates a specific protein or
proteins. Such phosphorylation reactions are known to be involved in the
opening of some calcium channels as well as in the activation of other enzymes.
In this system, the receptor is in the membrane with its binding site on the
outer surface. The G protein is totally within the membrane while the adenylyl
cyclase is within the mem-brane but projects into the interior of the cell. The
cAMP is generated within the cell (see Figure 10.4).
Whether or not a particular
agonist has any effect on a particular cell depends initially on the presence
or absence of the appropriate receptor. However, the na-ture of the response depends on these factors:
·
Which G protein couples with the receptor
·
Which kinase is activated
· Which proteins are accessible for the kinase to phosphorylate
The variety of possible
responses is further in-creased by the fact that receptor-coupled G proteins
can either activate enzymes other than adenylate cy-clase or can directly
influence ion channel functions.
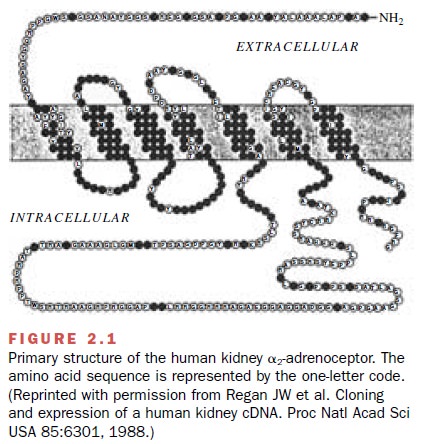
Many different receptor types
are coupled to G pro-teins, including receptors for norepinephrine and epi-nephrine
( α- and β-adrenoceptors),
5-hydroxytrypta-mine (serotonin or 5-HT receptors), and muscarinic
acetylcholine receptors. Figure 2.1 presents the struc-ture of one of these,
the α2-adrenoceptor from the hu-man
kidney. All members of this family of G protein–coupled receptors are
characterized by having seven membrane-enclosed domains plus extracellular and
intracellular loops. The specific binding
sites for ag-onists occur at the extracellular surface, while the inter-action
with G proteins occurs with the intracellular por-tions of the receptor. The
general term for any chain of events
initiated by receptor activation is signal
trans-duction.
Related Topics