Chapter: Essentials of Psychiatry: Psychiatric Pathophysiology: Schizophrenia
Postmortem Limbic Cortex Abnormalities in Schizophrenia: Structural Changes
Postmortem Limbic Cortex
Abnormalities in Schizophrenia: Structural Changes
Much focused work in the human limbic cortex in
schizophrenia began after Scheibel and Kovelman (1981) described an alteration
in pyramidal cell apical dendrite orientation in the hippocampus, in its
anterior and middle section, particularly at the subicular-CA1
border. In a later extension of this work, they correlated symptom severity
with the extent of the dendritic disorientation (Kovelman and Scheibel, 1984).
However, other studies have not uniformly replicated these findings (Altshuler et al., 1987; Christison et al., 1989; Vogel et al., 1997), but the original find-ings are still often
referenced as evidence for neuroanatomical abnormalities in the hippocampus.
Weinberger (1996) highlights several studies describing cytoarchitectural
abnormalities in the entorhinal cortex (including the specific loss of
NADPH-diapho-rase neurons Nicotinamide Adenosine Dinucleotide Phosphate) as providing
the best evidence for neuropathological findings (Akbarian et al., 1993; Arnold et al.,
1991; Jakob and Beckmann, 1986). Other studies have reported significant
reductions in the volumes or cross-sectional areas of the entorhinal cortex or
hip-pocampus in schizophrenia (Bogerts et
al., 1985; Brown et al., 1986;
Colter et al., 1987; Falkai and
Bogerts, 1986; Falkai et al., 1988),
but these results have not been uniformly replicated (Benes et al., 1991; Heckers et al., 1991).
Hippocampal size is reduced bilaterally, albeit
mildly, in the illness especially in anterior areas (Becker et al., 1996; Bilder et al., 1995; Bogerts et al., 1990; Suddath et al., 1989. Shape analyses of the
hippocampus have suggested regional abnormalities of volume in schizophrenia.
Importantly, regional shape abnormalities are predominantly localized to the
head, implicating only a delimited area within hippocampus as abnor-mal
(Csernansky et al., 1998).
Neurochemical Changes
Changes in GABAA receptor density, in
GABA release and in glutamate-related transmitters and their enzymes in
hippoc-ampus have been reported in the illness (Simpson et al., 1992; Tsai et al.,
1995). While there appears to be no change in the density of hippocampal NMDA glutamate
receptors (Ishimaru et al., 1992;
Kerwin et al., 1990; Kornhuber et al., 1989), kainate binding, particularly in CA2
has been found reduced in several studies (Kerwin et al., 1988, 1990; Simpson et
al., 1992) but not consistently (Deakin et
al., 1989). Reduced levels of non-NMDA receptor binding (Kerwin et al., 1990) and lower concentrations
of non-NMDA receptor mRNA (Harrison et al.,
1991), have both been reported in CA3. The previous finding of an
alteration in the NR1 subunit and an increase in NR2B in
postmortem tissue from schizophrenia (Gao et
al., 2000) suggests a reduction in excita-tory glutamate transmission at
hippocampal NMDA receptors in this illness.
In addition, considerable evidence of compromised
cogni-tive function, especially short-term memory and attention, exists in
schizophrenia (Green, 1996; Gruzelier et
al., 1988; Venables, 1992). These dysfunctions may represent the behavioral
corre-lates of hippocampal pathology.
In Vivo
Functional Limbic Cortex Change in Schizophrenia
Functional studies of human brain in schizophrenia
have directly demonstrated an alteration in neuronal activity in the limbic
cor-tex in the illness (Fletcher, 1998; Haznedar et al., 1997; Heckers et al.,
1998; Medoff et al., 2001; Nordahl et al., 1996; Tamminga et al., 1992). Anterior cingulate
cortex consistently shows altera-tions in schizophrenia when persons are imaged
medication-free and matched for performance. Moreover, connectivity analyses
suggest that the anterior cingulate rCBF is not tightly coupled to hippocampal
activity during tasks of learning and memory, as it is in normal persons.
Although the entire body of these data have not yet suggested the pivotal
limbic pathology, they do implicate abnormal function of these structures in
the illness. Moreover, as suggested in the preclinical studies, such pathology
could desta-bilize the function of subcortical brain areas in psychosis.
An analysis of connectivity in auditory
discrimination ex-periments directly suggests that a systems failure occurs
within limbic cortex in schizophrenia. A malfunction within the limbic cortex
and the subsequent disruption of related neocortical areas and secondary
dysregulation of subcortical structures may under-lie the manifestations of
schizophrenia. This working hypothesis suggests that it may be the resultant
“misbehavior” of the limbic system itself that generates positive and cognitive
symptoms in schizophrenia, possibly through its connections with neocortical
and subcortical structures. These speculations raise the possibil-ity that the
primary origin of the circuit dysfunction could be varied, but inevitably
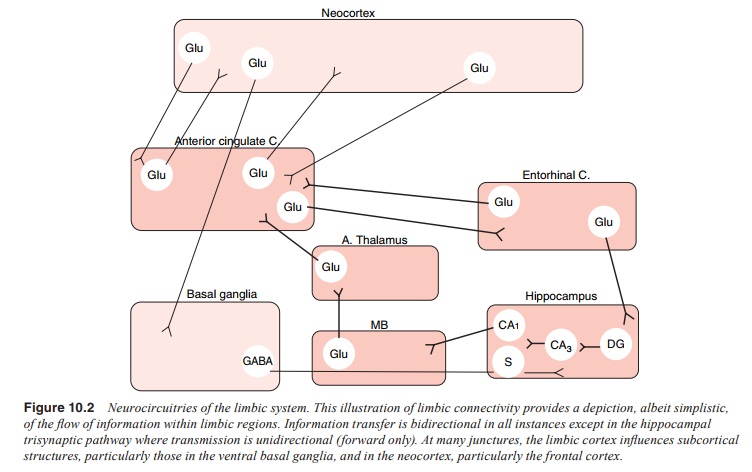
result in a characteristic “psychosis” circuit abnormality (Figure 10.2).
Functional Effects of D2 Dopamine Receptor Blockade
The clinical evidence of a systems basis of
psychosis, along with the preclinical data suggesting multiple and complex
neurotrans-mitter interactions within these symptoms, builds a plausible
“psychosis circuit” for schizophrenia and potentially for its treat-ment. The
effective functioning of the human brain to facilitate cognitive, motor and
affective performance, is dependent not only on the proper functioning of
individual regional neuronal groups dedicated to specific tasks, but also to
interacting brain systems which function to connect neural systems to perform a
particular mental task and systematically to direct information flow in the
brain. Because schizophrenia is not characterized pri-marily by a neural or
behavioral deficit, but rather by productive symptoms and by a “confusion” of
neural activity with result-ing mental malfunction, a system hypothesis of
schizophrenia pathology is plausible. The idea that psychosis is the
consequence of dysfunction somewhere within the limbic cortex, seen during
cognitive work and even during routine mental function is sup-ported by a great
deal of research. This putative dysfunction is prominently manifest in the
anterior components of the limbic system. Hence this change primarily
influences the frontal re-gions of the neocortex, leaving the posterior
hippocampus and the posterior neocortex relatively unaffected. Exactly where
the primary limbic pathology is located within these anterior areas is a matter
of speculation, but it could be multiple sites with a single resultant systems
dysfunction.
Importantly, this idea allows for the real possibility that other drug actions can be exerted at other sites within the relevant circuits. The 5HT2A antagonist activity of the second generation drugs may well be exerted, either partially or wholly, in frontal cortex. Moreover, the glutamate-enhancing activity of the second generation drugs at the NMDA receptor, whether tied to this sero-tonin action or independent from it, may well be a cortical action. This formulation increases the complexity but also the therapeutic potential of psychosis treatment(CarlssonandCarlsson,1990).
Related Topics