Chapter: Pathology: Gastrointestinal Tract Pathology
Pathology: Stomach
STOMACH
Congenital Disorders
Pyloric stenosis is a congenital stenosis of the pylorus due to
marked muscular hypertrophy of the pyloric
sphincter, resulting in gastric outlet obstruction. It affects male infants
more than females. It is associated with Turner and Edwards syn-dromes.
Presentation is the onset of regurgitation and vomiting in week 2 of life; waves
of peristalsis are visible on the abdomen and there is a palpable oval
abdomi-nal mass. Treatment is surgical.
Congenital diaphragmatic hernia occurs when a congenital
defect is present in the diaphragm, resulting in herniation of the
abdominal organs into the thoracic cav-ity. The stomach is the most commonly
herniated organ due to left-sided congenital diaphragmatic hernia. Congenital
diaphragmatic hernia is often associated with intestinal malrotation. It may be
complicated by significant lung hypoplasia.
Hypertrophic Gastropathy
Ménétrier disease is a rare disease of
middle-aged men. It is caused by profound hyperplasia of surface mucous
cells, accompanied by glandular atrophy. It is char-acterized by enlarged rugal
folds in the body and fundus; clinically, patients experi-ence decreased acid
production, protein-losing enteropathy, and increased risk of gastric cancer.
Zollinger-Ellison syndrome.
Acute Inflammation and Stress Ulcers
Acute hemorrhagic gastritis causes acute inflammation,
erosion, and hemorrhage of the gastric mucosa,
secondary to a breakdown of the mucosal barrier and acid-induced injury. The
etiology is diverse, with initiating agents including chronic aspirin or NSAID
use, alcohol use, smoking, recent surgery, burns, ischemia, stress, uremia, and
chemotherapy. Patients present with epigastric abdominal pain, or with gastric
hemorrhage, hematemesis, and melena.
Gastric stress ulcers are multiple, small, round,
superficial ulcers of the stomach and duodenum. Predisposing
factors include:
•
NSAID use
•
Severe stress
•
Sepsis
•
Shock
•
Severe burn or trauma
•
Elevated intracranial pressure (Cushing ulcers)
ICU patients have a high
incidence of gastric stress ulcer. These ulcers may be com-plicated by
bleeding.
Chronic Gastritis
Chronic gastritis is chronic
inflammation of the gastric mucosa, eventually leading to atrophy (chronic
atrophic gastritis).
Fundic type chronic gastritis is an autoimmune atrophic
gastritis that involves the body and the fundus. It is
caused by autoantibodies directed against parietal cells and/or intrinsic
factor. The result is loss of parietal cells, decreased acid secretion,
increased serum gastrin (G-cell hyperplasia), and pernicious anemia
(megaloblastic anemia due to lack of intrinsic factor and B12 malabsorption).
Women are affected more than men.
Grossly, one sees a loss of
rugal folds in the body and fundus. Microscopically, mucosal atrophy is seen
with loss of glands and parietal cells, chronic lymphoplas-macytic
inflammation, and intestinal metaplasia. Patients are at increased risk for
gastric carcinoma.
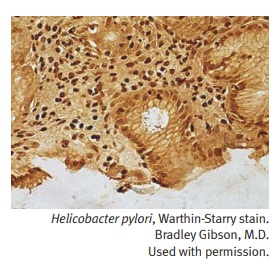
Antral type chronic gastritis (also called Helicobacter pylori gastritis) is the most common form of chronic
gastritis in the United States. The H.
pylori organisms are curved, gram-negative rods which produce urease. The
risk of infection increases with age. Infection is also associated with duodenal/gastric
peptic ulcer, and gastric carcinoma with intestinal type histology.
Microscopically, H. pylori organisms are visible in the
mucous layer of the surface epithelium. Other microscopic features include foci
of acute inflammation, chronic inflammation with lymphoid follicles, and
intestinal metaplasia.
Chronic Peptic Ulcer (Benign Ulcer)
Peptic ulcers are ulcers of the distal
stomach and proximal duodenum caused by gastric secretions
(hydrochloric acid and pepsin) and impaired mucosal defenses. Predisposing
factors include the following:
•
Chronic NSAID and aspirin use
•
Steroid use
•
Smoking
•
H. pylori infection
Patients present with burning
epigastric pain. Diagnosis is by endoscopy with or without biopsy. Treatment is
acid suppression (H2 blocker, proton pump inhibitor, etc.) and eradication of H. pylori.
Complications of peptic ulcer
include hemorrhage, iron deficiency anemia, penetra-tion into adjacent organs,
perforation (x-ray shows free air under the diaphragm), and pyloric
obstruction.
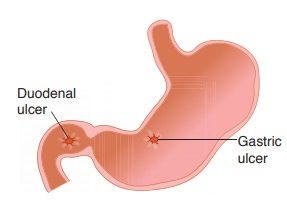
Duodenal peptic ulcers are more common than gastric
peptic ulcers. Associations include the following:
•
H. pylori (~100%)
•
Increased gastric acid secretion
•
Increased rate of gastric emptying
•
Blood group O
•
Multiple endocrine neoplasia (MEN) type I
•
Zollinger-Ellison syndrome
•
Cirrhosis
•
Chronic obstructive pulmonary disease
Most duodenal peptic ulcers
are located in the anterior wall of the proximal duo-denum.
Gastric peptic ulcers are associated with H. pylori (75%). Most are located in the lesser curvature of the antrum. Grossly, they
are small (<3 cm), sharply demar-cated (‘punched out’), solitary with
round/oval shape, smooth borders, and radiating mucosal folds.
Gastric Carcinoma (Malignant Ulcer)
Gastric carcinoma is more
common in Japan than in the United States, and has a decreasing incidence in
the United States. Dietary factors can be risk factors:
•
Smoked fish and meats
•
Pickled vegetables
•
Nitrosamines
•
Benzpyrene
•
Reduced intake of fruits and vegetables
Other risk factors include H. pylori infection, chronic atrophic gastritis, smoking, blood
type A, bacterial overgrowth in the stomach, prior subtotal gastrectomy, and
Ménétrier disease.
Gastric carcinoma is often (90%) asymptomatic
until late in the course, when it can produce weight loss and anorexia. It can
also present with epigastric abdominal pain mimicking a peptic ulcer, early
satiety, and occult bleeding with iron deficiency anemia.
Most gastric carcinomas are located in the
lesser curvature of the antrum. They are large (>3 cm) ulcers with heaped-up
margins and a necrotic ulcer base. They may also occur as a flat or polypoid
mass. Several histological types occur.
•
The intestinal type
shows gland-forming adenocarcinoma microscopically.
•
The diffuse type shows
diffuse infiltration of stomach by poorly differenti-ated tumor cells, numerous
signet-ring cells (whose nuclei are displaced to the periphery by intracellular
mucin), and linitis plastica
(thickened “leather bottle”–like stomach) gross appearance.
Gastric carcinoma may specifically metastasize
to the left supraclavicular lymph node (Virchow sentinel node) and to the ovary
(Krukenberg tumor).
Diagnosis is by endoscopy
with biopsy; treatment is gastrectomy. The prognosis is poor, with overall
5-year survival ~30%.
Related Topics