Chapter: Microbiology and Immunology: Bacteriology: Yersinia
Pathogenesis and Immunity - Yersinia pestis
Pathogenesis and Immunity
Y. pestis is a highly virulent bacterium, which causes plague witha high mortality rate. The ability of Yersinia species to resist phagocytic killing is the hallmark of pathogenesis of plague. Serum resistance and the ability of the bacilli to absorb organic iron as a result of a siderophore-independent mechanism fur-ther contributes to the pathogenesis of the disease.
◗ Virulence factors
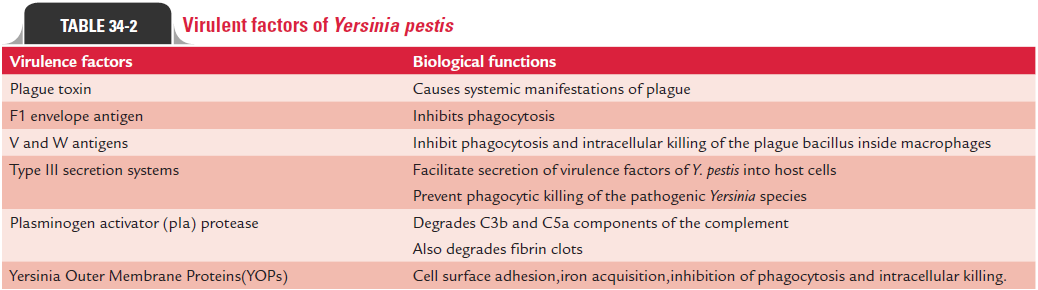
Y. pestis produces many toxins and enzymes, all of which con-tribute to pathogenesis of the diseases. Virulence factors of Y. pestislike that of Salmonella are complex. These are encodedboth on the organism’s chromosome and on large plasmids (Table 34-2).
Plague toxin: The term “plague toxins” refers to a complexof at least two different types of toxins: (a) endotoxin and (b) murine toxin, found in culture filtrates or in cell lysates.
A. The endotoxin is a lipopolysaccharide found in the cell wall and is responsible for many of the systemic manifesta-tions of the disease caused by Y. pestis.
B. Murine toxins exhibit some properties of both exotoxins and endotoxins.
· It is a heat-labile protein. The toxins may be toxoided but do not diffuse freely into the medium and are released only by the lysis of the cell.
· The toxin is pathogenic in rats and mice but not in guinea pigs and rabbits and in humans and other pri-mates. The murine toxins produce local edema and necrosis with systemic effects on the peripheral vas-cular system and liver in experimental animal infec-tions. The role of murine toxins in the pathogenesis of plague in humans is not known.
F1 envelope antigen: F1 envelope antigen is a major virulencefactor, which inhibits phagocytosis and plays an important role in conferring protective immunity in humans and in mice.
V and W antigens: These two antigens inhibit phagocytosisand intracellular killing of the bacillus inside macrophages.
Type III secretion systems: Type III secretion systems (TTSS)consist of many proteins, which facilitate secretion of virulence factors of Y. pestis into host cells. The ability of the pathogenic Yersinia species to resist phagocytic killing is mediated by theTTSS. The bacteria on coming in contact with phagocytic cells in infected hosts secrete a variety of proteins, which prevent phago-cytic killing of the bacteria. Firstly, the proteins secreted into the phagocyte dephosphorylate several proteins required for phago-cytosis (YopH gene product) and then induce cytotoxicity by disrupting actin filaments (YopE gene product). Subsequently, proteins initiate apoptosis in macrophages (YopJ/P gene prod-uct). The TTSS also inhibits production of cytokines, which in turn reduces the inflammatory immune response to infection.
Other virulence factors: Yersiniaproduces the enzymes, suchas coagulase, fibrinolysin, and plasminogen activator (pIa) protease, which contributes to virulence of the bacterium. For example, enzyme plasminogen activator (pIa) protease degrades C3b and C5a components of the complement, thereby prevent-ing opsonization and phagocytic migration, respectively. The enzyme also degrades fibrin clots, thereby facilitating Y. pestis to spread rapidly. The virulence of the bacterium is enhanced further by its ability to absorb organic iron as a result of a sid-erophore-independent mechanism.
◗ Pathogenesis of plague
Plague in humans can occur either by the bite of a vector, by close contact with infected tissue or body fluids, or by direct inhalation of the bacterium.
Bubonic plague: It is a zoonotic disease transmitted by ratflea Xenopsylla cheopis from animals to humans. Y. pestis is a natural parasite of rodents; infection is transmitted and main-tained among them by rat fleas. The fleas acquire the infection by feeding on infected rodents. When rat flea bites an infected and diseased rat, it sucks nearly 0.5 mL of blood per feed con-taining nearly 5000–50,000 plague bacilli. In the stomach of the flea, the bacilli multiply to such an extent that they block the proventriculus and thereby prevent food entry into the stomach. The interval between the ingestion of infected blood and blocking in the proventriculus is referred to as extrinsic incubation period, which varies from 2 to 3 weeks in X. cheopis.
The blocked flea, to overcome starvation, begins a blood-suck-ing rampage by biting other rodents. Between its attempts to swal-low, the distended bacillus-packed stomach recoils, depositing the bacilli into the victim’s skin, thereby transmitting the infection. When the diseased rat dies, the fleas leave the carcass and in the absence of another rat, may bite human, causing bubonic plague.
The bubonic plague is characterized by the pathognomonic “bubo”. The disease occurs following the bite of rat flea, depos-iting thousands of bacilli into the host’s skin during blood meals. The bacilli migrate to the regional lymph nodes, where they are phagocytosed by the polymorphonuclear cells and mononuclear phagocytes, and multiply intracellularly.
The bacilli invade the nearby lymphoid tissue producing the characteristic—an inflamed, necrotic, and hemorrhagic—lymph node known as bubo (bubon meaning groin), as demonstrated in bubonic plague. Infected lymph nodes show large number of plague bacilli, destruction of the normal architecture, and medullary necrosis.
Subsequent spread of bacteria occurs along the lymphatic channels towards the thoracic duct, with eventual distribution of the bacteria in circulation leading to bacteremia and septice-mia. The bacteria, in the absence of specific therapy, potentially infect every organ, including the lungs, liver, spleen, kidneys, and rarely even the meninges.
Pneumonic plague: It is transmitted from humans tohumans. It occurs following direct inhalation of the bacilli by droplet infection due to close contact with infected hosts or by inhalation of aerosolized bacteria, such as may occur if used as a biological weapon. The bacilli spread through the lungs, producing a severe and rapidly progressive multilobar bron-chopneumonia, subsequently leading to bacteremia and septi-cemia. It is not a zoonotic disease.
Septicemic plague: It is usually the terminal stage of thebubonic or pneumonic plague. It may sometimes occur pri-marily. Primary septicemic plague may occur when the plague bacilli are deposited directly in the circulation, bypassing the lymphatics. Early dissemination with sepsis occurs, but without the formation of a bubo. This usually is observed in individuals bitten by the rat flea in the oral, tonsillar, and pharyngeal area and is due to the vascularity of the tissue and short lymphatic distance to the thoracic duct.
◗ Host immunity
An attack of plague provides a long-lasting immunity to infected humans.
Related Topics