Chapter: Medicine and surgery: Overdose, poisoning and addiction
Paracetamol poisoning - Overdose and poisoning
Paracetamol poisoning
Definition
Accidental or deliberate overdose of paracetamol, causing liver damage.
Incidence
Currently the commonest drug used for deliberate over-dose.
Aetiology
Overdose is generally accidental in toddlers. In older patients it is usually a form of deliberate self-harm; however, it may be accidental due to combination drug use.
Pathophysiology
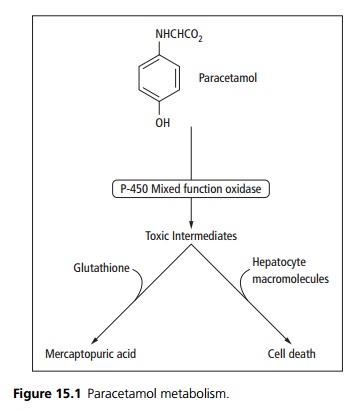
Paracetamol in overdose causes hepatocellular necrosis. Normally toxic metabolites are inactivated by conjugation with glutathione. When glutathione and other conjugating substances become depleted, the metabolites (e.g. epoxides) bind to intracellular proteins and cause hepatocyte damage (see Fig. 15.1). Liver failure leads to encephalopathy, haemorrhage, hypoglycaemia, cerebral oedema and death. Paracetamol can also cause renal failure.
Clinical features
There are often no early symptoms following paracetamol overdose, patients may present with nausea, vomiting and pallor, which usually settle within 24 hours. Right subcostal pain and tenderness may then develop, and after 48 hours jaundice and a large, tender liver are apparent, indicating hepatic necrosis. Other features include hypotension, arrhythmias, excitement, delirium and coma.
Investigations
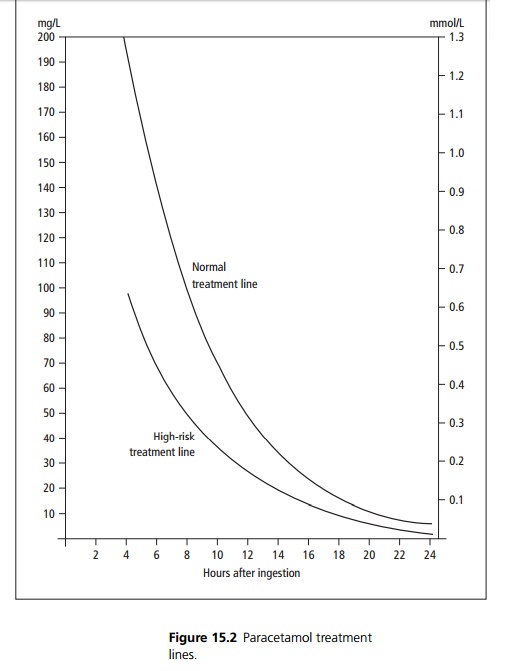
Serum paracetamol levels are taken 4 hours after ingestion and plotted to demonstrate if treatment is required (see Fig. 15.2). Patients on liver enzyme inducing drugs, malnourished patients or those who consume alcohol above recommended limits require treatment if levels are above the high-risk treatment line. In significant over-dose a prothrombin time, liver and renal function tests and a lactate should be checked and repeated at 24 hours. Prothrombin time measured 24 hours post ingestion is the best marker for liver damage.
Management
Activated charcoal is given if the patient presents within 1 hour of ingestion and >12 g (6 g in the high
risk treatment group) or ≥150 mg/kg have been ingested, whichever is the smaller.
N-acetylcysteine (a glutathione precursor) is given by intravenous infusion if the plasma paracetamol level is higher than the appropriate treatment line. It is maximally effective before 8 hours following ingestion but may be of value up to and beyond 24 hours. It may be appropriate to start N-acetylcysteine prior to blood levels are known if very high doses have been taken or if presentation is delayed.
Severe hepatotoxicity may necessitate liver transplantation.
Prognosis
If acute hepatic failure occurs, mortality is <50% with good management.
Related Topics