Chapter: Modern Pharmacology with Clinical Applications: β-Lactam Antibiotics
Monobactams
MONOBACTAMS
Another interesting group of
compounds produced by several bacterial genera are the monocyclic β-lactams (monobactams).
The natural monobactams have little antimicrobial activity. A synthetic
derivative, aztreonam (Azactam), has
excellent activity against gram-negative organisms, including P. aeruginosa. Aztreonam has low
affinity for penicillin-binding proteins in streptococci, staphylococci, and
anaerobes and therefore has no sig-nificant activity against gram-positive
bacteria or anaer-obes. Specific activity against gram-negative organisms
relates to the aminothiazolyl oxime moiety on the acyl side chain. Addition of
two methyl groups and a car-boxylic acid group on the oxime side chain enhances
ac-tivity against P. aeruginosa.
Aztreonam is stable to most β-lactamases (chromosomal and plasmid).
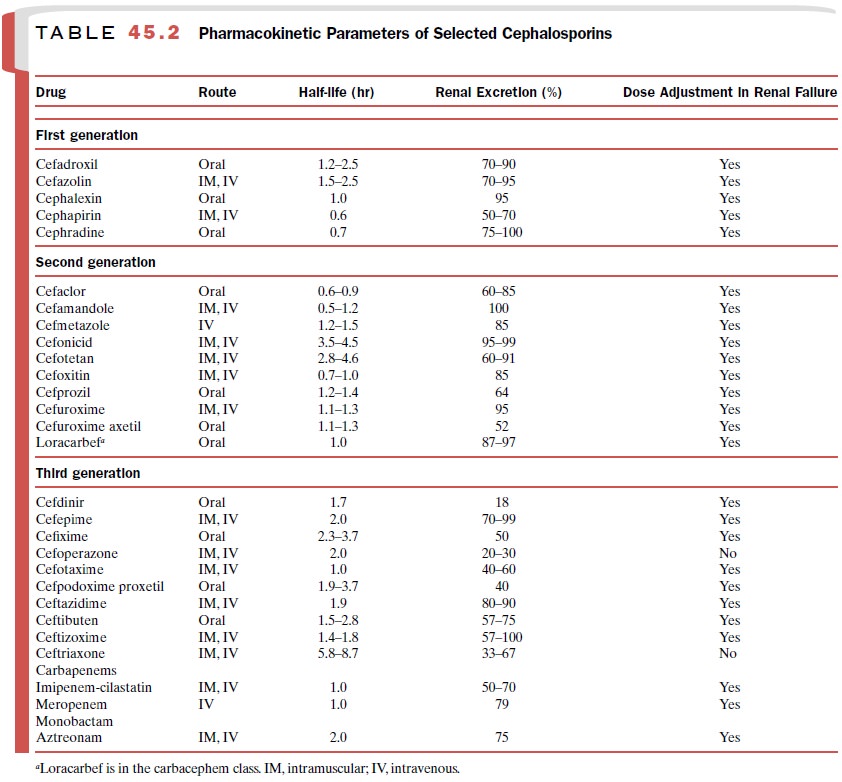
The pharmacokinetic
properties of aztreonam are similar to those of the parenteral cephalosporins
(Table 45.2). Aztreonam is not bioavailable after oral adminis-tration. During
its distribution phase, the drug can achieve therapeutic concentrations in cerebrospinal
fluid in the presence of inflamed meninges. Conse-quently, aztreonam is an
alternative antibiotic to the cephalosporins for the therapy of meningitis
caused by gram-negative bacilli.
Aztreonam may be used as a
substitute for an aminoglycoside in the treatment of infections caused by
susceptible gram-negative organisms. Most of the ad-verse effects of aztreonam
are local reactions at the site of injection. Interestingly, aztreonam rarely
causes al-lergic reactions in patients with a history of type I
hy-persensitivity to other β-lactam antibiotics.
Related Topics