Chapter: Microbiology and Immunology: Culture Methods
Methods of Culture
Methods of Culture
Various methods are used for culturing of bacteria. These include (a) streak culture, (b) lawn culture, (c)
pour-plate culture, (d ) stroke
culture, (e) stab culture, and ( f ) liquid culture.
Streak Culture
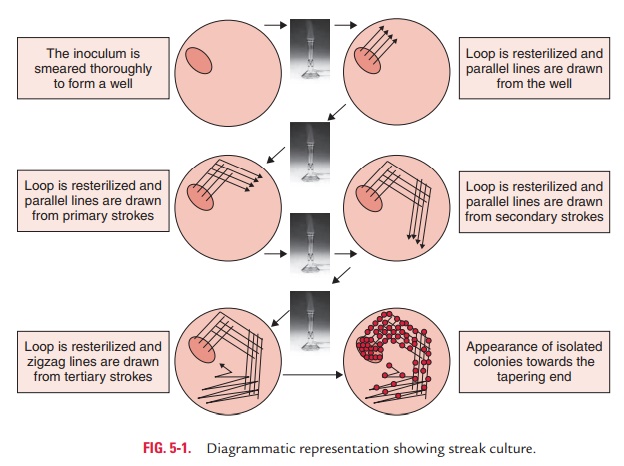
Streak culture is the most useful method for obtaining discrete colonies
of the bacteria. It is carried out by streaking on the surface of a solid media
plate using a platinum or nichrome loop of 2–4 mm diameter. In this method, a
loopful of the inoculum is placed near the peripheral area of the plate. The
inoculum is then spread with the loop to about one-fourth of the plate with
close parallel strokes. From the primary inoculum, it is spread thinly over the
plate by streaking with the loop in parallel lines. The loop is flamed and
cooled in between the streaks to obtain isolated colonies. The inoculated
culture plate is incubated at 37°C overnight for demonstration of colonies.
Confluent growth occurs at the primary inoculum, but becomes progressively
thinner, and well-separated colonies are demonstrated on the final streaks of
the inoculum (Fig. 5-1). Single isolated colonies obtained by this method are
very useful to study various properties of bacteria. Streak culture is the most
useful method for obtaining discrete colonies of the bacteria.
Lawn Culture
The lawn culture provides a uniform layer of bacterial growth on a
solid medium. It is carried out by flooding the surface of the solid media
plate with a liquid culture or suspension of bacteria, pipetting off the excess
inoculum, and finally incubating the plate overnight at 37°C. Alternatively,
the cul-ture plate may be inoculated by a sterile swab soaked in liquid
bacterial culture or suspension and incubating overnight for demonstration of
the bacterial colonies.
Pour-Plate Culture
The pour-plate culture is used to determine approximate number of
viable organisms in liquids, such as water or urine. It is used to quantitate
bacteria in urine cultures and also to estimate the viable bacterial count in a
suspension. This method is carried out in tubes, each containing 15 mL of
molten agar. The molten agar in tubes is left to cool in a water bath at 45°C.
The inoculum to be tested is diluted in serial dilution. Then 1 mL each of
diluted inoculum is added to each tube of molten agar and mixed well. The
contents of tubes are poured into sterile Petri dishes and allowed to set.
After overnight incubation of these Petri dishes at 37°C, colonies are found to
be distributed throughout the depth of the medium, which can be counted using a
colony counter.
Stroke Culture
Stroke culture provides a pure growth of bacteria for carrying out
slide agglutination and other diagnostic tests. It is carried out in tubes
usually containing nutrient agar slopes.
Stab Culture
Stab culture is prepared by stabbing the medium in tubes with a
long, straight wire and incubating at 37°C.
Liquid Culture
Liquid culture is prepared in a liquid media enclosed in tubes,
flasks, or bottles. The medium is inoculated by touching with a charged loop or
by adding the inoculum with pipettes or syringes and incubating at 37°C,
followed by subculture on to solid media for final identification.
A major disadvantage of liquid culture is that it does not provide
pure culture of the bacteria and also the bacterial growth does not exhibit
special characteristic appearances.
Anaerobic Culture
Obligate anaerobes are bacteria that can live only in the absence
of oxygen. These anaerobes are killed when exposed to the atmosphere for as
briefly as 10 minutes. Some anaer-obes are tolerant to small amounts of oxygen.
Facultative anaerobes are those anaerobes that grow with or without oxygen.
Anaerobic bacterial culture is a method used to grow anaer-obes
from a clinical specimen. Culture and identification of anaerobes is essential
for initiating appropriate treatment.
The failure to do so may have serious consequences, such as
amputation, organ failure, sepsis, meningitis, and even death.
Related Topics