Chapter: Modern Analytical Chemistry: Calibrations, Standardizations, and Blank Corrections
Linear Regression and Calibration Curves
Linear Regression
and Calibration Curves
In a single-point external standardization, we first determine
the value of k by
measuring the signal for a single standard
containing a known
concentration of analyte. This value of k and the signal for the sample
are then used to calculate the concentration of analyte
in the sample (see Example
5.2). With only a single
determination of k, a
quantitative analysis using a single-point external stan- dardization is straightforward. This is also true for a single-point standard addi- tion (see
Examples 5.4 and
5.5) and a single-point internal standardization (see
Example 5.8).
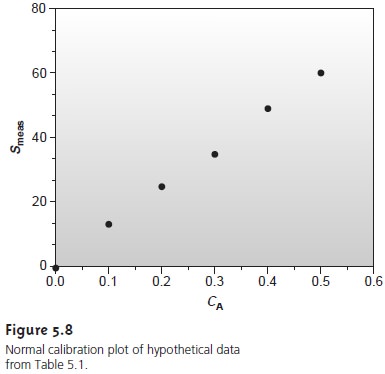
A multiple-point standardization presents a more
difficult problem. Consider the data in Table 5.1 for a multiple-point external
standardiza- tion. What is the best estimate of the relationship
between Smeas and CS? It is tempting to treat
this data as five separate single-point standardizations, determining k for each stan- dard and reporting the mean value.
Despite its simplicity, single-point
standardizations, determining k for
each stan dard and reporting the mean value.
Despite its simplicity, this is not an appropriate
way to treat a multiple-point standardization.
In a single-point standardization, we assume that the reagent
blank (the first
row in Table
5.1) corrects for
all constant sources of determinate error. If this is not the case, then the
value of k determined by a single- point standardization will have a determinate error.
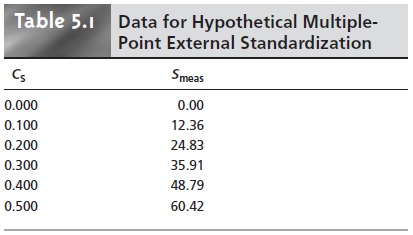
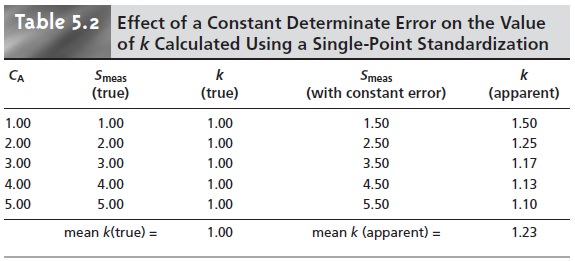
Table 5.2 demonstrates how an uncorrected constant error affects
our determination of k. The first
three columns show the
concentration of analyte,
the true measured
signal (no constant error)
and the true
value of k for
five standards. As expected, the value of k is the same for each standard. In the fourth column a constant determinate error of +0.50 has been
added to the measured signals. The corresponding val- ues
of k are shown
in the last column. Note that a different
value of k is obtained for
each standard and
that all values
are greater than the
true value. As we noted,
this is a significant limitation to any single-point standardization.
How do we find the best estimate
for the relationship be- tween the measured signal and the
concentration of analyte
in a multiple-point standardization? Figure 5.8 shows
the data in Table
5.1 plotted as a normal calibration curve. Although the data
appear to fall along a straight line,
the actual calibration curve is not intuitively obvious. The process
of mathemati- cally determining the best equation for the calibration curve is called regression.
Linear Regression of Straight-Line Calibration Curves
A calibration curve shows us the relationship between the measured
signal and the analyte’s concentration in a series of standards. The most useful
calibration curve is a straight line since the method’s sensitivity is the same for all concentrations of an-
alyte. The equation for a linear calibration curve is
y = β0 + β1x
….5.12
where y is the signal and x is the amount of analyte. The constants β0 and β1 are the
true y-intercept and the true slope,
respectively. The goal of linear regres- sion is to determine the best estimates for the slope,
b1, and
y-intercept, b0. This is
accomplished by minimizing the residual
error between the experimental val- ues, yi, and those values,
yˆi, predicted by equation 5.12 (Figure 5.9).
For obvious reasons, a regression analysis
is also called a least-squares treatment. Several ap- proaches to the linear
regression of equation 5.12 are discussed in the following sections.

Unweighted Linear Regression with Errors in y
The most commonly
used form of linear regression is based on three assump- tions: (1) that any difference between
the experimental data and the calculated
regression line is due to indeterminate errors affecting the values of y, (2) that these indeterminate errors are
normally distributed, and
(3) that the
indetermi- nate errors in y do not depend on the value
of x. Because we assume that indeter-
minate errors are the same for all standards, each standard contributes equally in
estimating the slope
and y-intercept. For this reason the result is considered an unweighted linear regression.
The second assumption is generally true because of the central
limit theorem. The validity of the two remaining assumptions is less cer- tain and should be evaluated before
accepting the results
of a linear regression.
In particular, the
first assumption is always suspect
since there will
certainly be some indeterminate errors affecting the values of x. In preparing
a calibration curve, however, it is not
unusual for the
relative standard deviation of the mea- sured signal (y) to be significantly larger
than that for
the concentration of ana-
lyte in the standards (x). In such circumstances, the first assumption is usually
reasonable.
Finding the Estimated Slope and y-Intercept
The derivation of equations for cal-
culating the estimated slope and y-intercept can be found
in standard statistical texts7 and is not developed here. The resulting equation for the slope is given as
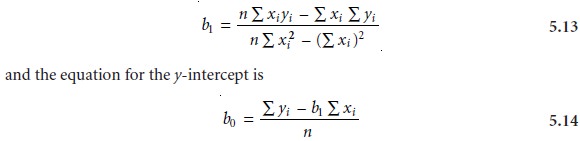
Although equations 5.13 and 5.14 appear formidable, it is only necessary to evaluate
four summation terms.
In addition, many
calculators, spreadsheets, and
other com- puter software packages are capable
of performing a linear regression analysis based on this
model. To save
time and to avoid tedious
calculations, learn how
to use one of
these tools. For
illustrative purposes, the
necessary calculations are
shown in de- tail in the following
example.

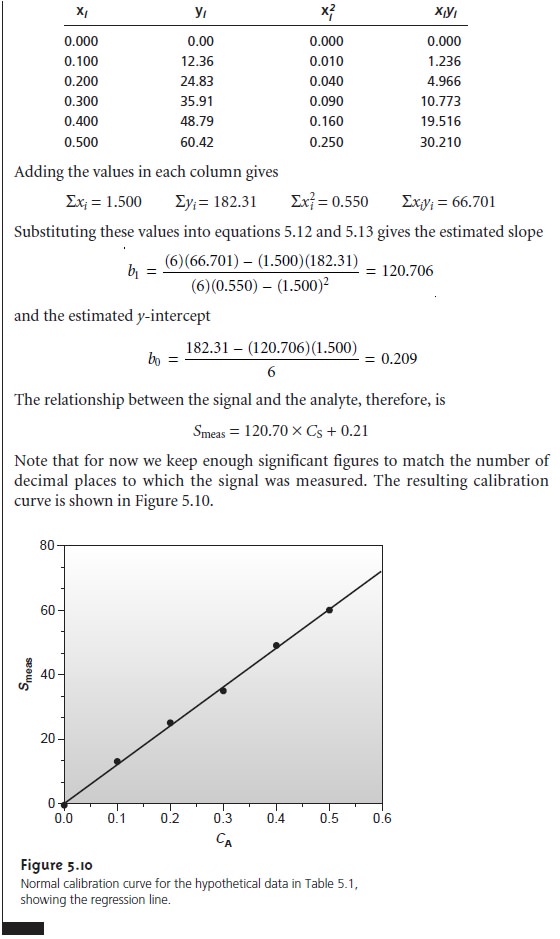
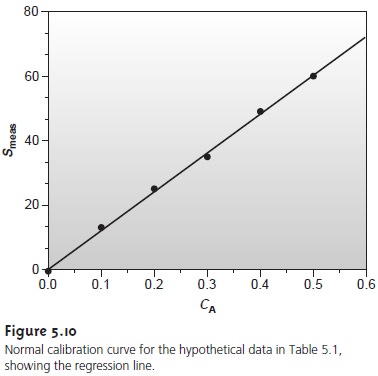
Uncertainty in the Regression Analysis
As shown in Figure 5.10, the regression line need not pass through the data points (this is the consequence of indeter- minate errors affecting the signal). The cumulative deviation of the data from the regression line is used to calculate the uncertainty in the regression due to indeterminate error.
This is called the standard deviation about the regression, sr,
and is given as

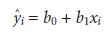
There is an obvious similarity between equation 5.15,
except that the sum of squares term for sr is determined rela- tive
to yˆi
instead of yˆ, and the denominator is n – 2 instead of n – 1; n – 2 indicates
that the linear
regression analysis has only n –
2 degrees of freedom since
two pa- rameters, the slope and the intercept, are used to calculate the values of yˆi.
A more useful
representation of uncertainty is to consider
the effect of indeter-
minate errors on the predicted slope and intercept. The standard deviation of the slope and intercept are given as
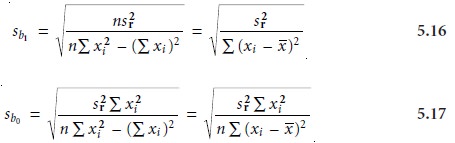

where t is selected
for a significance level of α and for n –
2 degrees of freedom. Note that the terms tsb1 and tsb0 do not contain a factor of (Root of n
)–1 because the con-fidence interval is based on a single
regression line. Again, many calculators, spread- sheets, and computer
software packages can handle the calculation of sb0 and
sb1 and the corresponding confidence intervals
for β0 and β1. Example
5.11 illustrates the calculations.


To minimize the uncertainty in the predicted slope and y-intercept, calibration curves are best prepared
by selecting standards that are evenly
spaced over a wide
range of concentrations or amounts
of analyte. The reason for this can be rational- ized by examining equations 5.16 and 5.17.
For example, both
sb0 and sb1 can be minimized by increasing the value of the term Σ(xi –
x ‘ )2, which
is present in the de-
|
i |
Using the Regression Equation
Once the regression equation is known,
we can use it
to determine the concentration of analyte in a sample.
When using a normal cali- bration curve with external
standards or an internal standards calibration curve, we
|
YX |
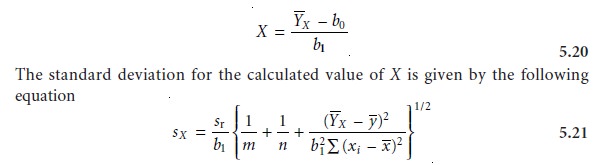
where m is the number of replicate samples
used to establish Y’X, n is
the number of calibration standards, yˆ is
the average signal for the standards, and xi and–x’ are
the individual and mean concentrations of the standards. Once sX is
known the confi- dence interval for the analyte’s concentration can be calculated as
μX= X ± tsX
where μX is the expected
value of X in
the absence of determinate errors,
and the value of t is determined by the desired
level of confidence and for n –
2 degrees of freedom. The following example
illustrates the use
of these equations for an analysis using a normal calibration curve with external
standards.


In a standard addition the analyte’s concentration is determined
by extrapolating the calibration curve to find the x-intercept. In this case
the value of X is
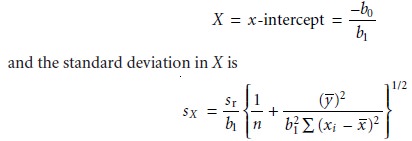
where n is the number of standards used in preparing
the standard additions
cali- bration curve (including the sample with no added
standard), and –y is
the average signal for the
n
standards. Because the
analyte’s concentration is determined by ex-
trapolation, rather than by interpolation, sX
for the method of standard additions
generally is larger than for
a normal calibration curve.
A linear regression analysis should not
be accepted without
evaluating the validity of the model
on which the
calculations were based.
Perhaps the simplest way to evaluate
a regression analysis
is to calculate and plot the residual error for each
value of x. The
residual error for
a single calibration standard, ri, is given as
ri = yi – yˆi
If the regression model is valid,
then the residual
errors should be randomly distrib- uted about an average
residual error of 0, with
no apparent trend
toward either smaller or larger residual
errors (Figure 5.11a).
Trends such as those shown
in Fig- ures 5.11b
and 5.11c provide
evidence that at least one of the assumptions on which
the regression model
is based are incorrect. For example, the trend toward
larger residual errors in Figure 5.11b
suggests that the
indeterminate errors affecting y are not independent of the value
of x. In Figure
5.11c the residual errors are not
ran- domly distributed, suggesting that the data cannot be modeled with a straight-line relationship. Regression methods for these two cases are discussed
in the following sections.

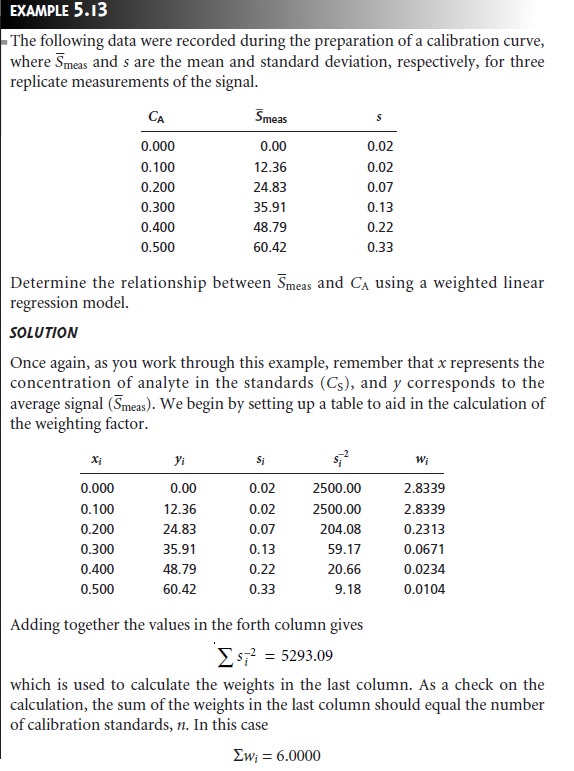
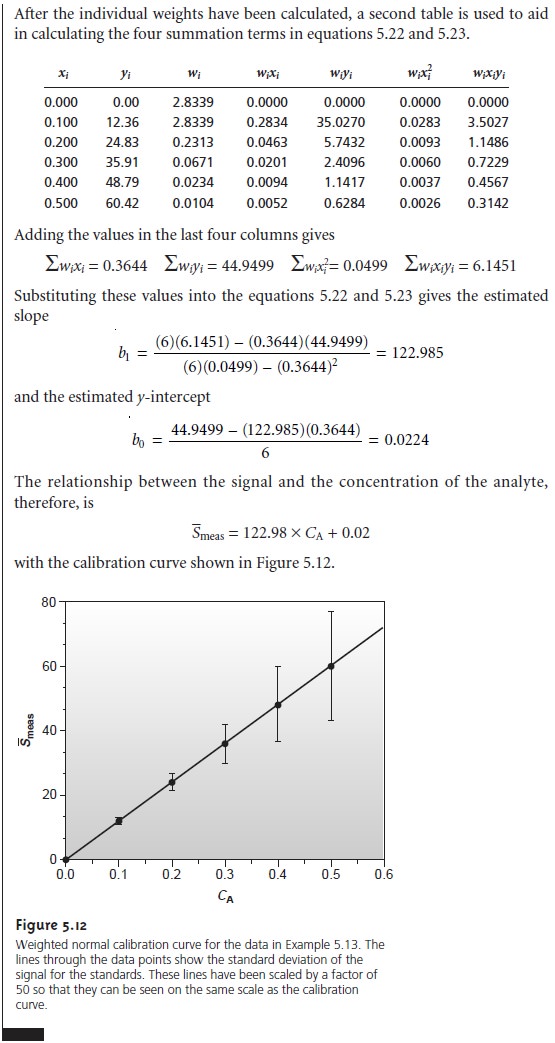
Weighted Linear Regression with Errors in y
Equations 5.13 for the slope, b1, and 5.14 for the y-intercept, b0, assume that
indeterminate errors equally
affect each value
of y. When this
assumption is false,
as shown in Figure 5.11b, the variance associated with each value of y must be included
when estimating β0 and β1. In this case the predicted slope and intercept are
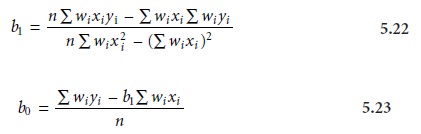
where wi is
a weighting factor accounting for the variance in measuring yi. Values of
wi are calculated using equation 5.24

where si is the standard deviation associated with
yi. The
use of a weighting factor ensures that the contribution of each pair of xy values to the regression line is proportional to the precision with which yi is
measured.
Equations for calculating confidence intervals for the slope, the
y-intercept, and the concentration of analyte when using a weighted
linear regression are not as easily defined
as for an unweighted
linear regression.
The confidence in- terval for the concentration of an analyte, however, will be at its optimum
valuewhen the analyte’s
signal is near the weighted centroid, –y, of the calibration curve
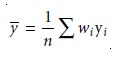
Weighted Linear Regression with Errors in Both x and y
If we remove
the assumption that the indeterminate errors affecting a calibration
curve are found only in the signal (y), then indeterminate errors affecting the preparation of standards containing known amounts of analyte (x) must
be fac- tored into
the regression model.
The solution for
the resulting regression line is
computationally more involved than that for
either the unweighted or weighted
regression lines, and is not presented in this text.
Curvilinear and Multivariate Regression
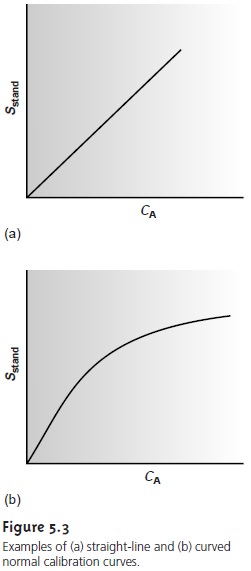
Regression models based on a straight line, despite their apparent complexity, use the simplest functional relationship between two variables. In many cases,
calibra- tion curves show a pronounced curvature at high concentrations of analyte (see Figure 5.3b). One approach to constructing a calibration curve
when curvature exists
is to seek a transformation function
that will make the data linear. Logarithms, expo- nentials, reciprocals, square
roots, and trigonometric functions have all been used in
this capacity. A plot of y versus log x is a typical example.
Such transformations are not
without complications. Perhaps
the most obvious
is that data
that originally has a uniform variance for the y values will not maintain that uniform variance when the variable is transformed.
A more rigorous
approach to developing a regression model
for a nonlinear calibration curve is to fit
a polynomial equation such as y =
a
+ bx + cx2 to the data.
Equations for calculating the parameters a, b, and c are
derived in the
same manner as that described earlier
for the straight-line model. When a single
polynomial equation cannot be fitted to the calibration data, it may be possible
to fit separate polynomial equations to short segments
of the calibration curve. The result is a sin- gle
continuous calibration curve
known as a spline function.
The regression models considered earlier
apply only to functions containing a single independent variable. Analytical methods, however, are frequently subject
to determinate sources of error due to interferents that contribute to the measured
sig- nal. In the
presence of a single interferent, equations 5.1 and
5.2 become
Smeas = kAnA + kInI + Sreag
Smeas = kACA + kICI + Sreag
where kI is the interferent’s sensitivity, nI is the moles
of interferent, and
CI is the in- terferent’s concentration.
Multivariate calibration curves can be prepared using standards that contain known
amounts of analyte
and interferent.
Related Topics