Chapter: Medical Microbiology: An Introduction to Infectious Diseases: Intravascular Infections, Bacteremia, and Endotoxemia
Intravascular Infection
INTRAVASCULAR INFECTION
Intracardiac infections (endocarditis) and those primarily involving veins (thrombophlebitis) or arteries (endarteritis) are usually caused by bacteria, although other agents including fungi and viruses have been occasionally implicated. This discussion will focus primarily on the bacterial causes, because they are the most frequent. Infections of the cardiovascular system are usually extremely serious and, if not promptly and adequately treated, can be fatal. They commonly produce a constant shedding of organisms into the bloodstream that is often characterized by continuous, low-grade bacteremia (1 to 20organisms/mL of blood) in untreated patients.
Infective Endocarditis
The term infective endocarditis is preferable to the commonly used term bacterial endocarditis, simply because not all infections of the endocardial surface of the heart are caused by bacteria. Most infections occur on natural or prosthetic cardiac valves, but can also develop on septal defects, shunts (eg, patent ductus arteriosus), or the mural endocardium. Infections involving coarctation of the aorta are also classified as infective endocarditis because the clinical manifestations and complications are similar.
Pathogenesis
The pathogenesis of infective endocarditis involves several factors that, if concurrent, re-sult in infection:
1. The endothelium is altered to facilitate colonization by bacteria and deposition of platelets and fibrin. Most infections involve the mitral or aortic valves, which are par-ticularly vulnerable when abnormalities such as valvular insufficiency, stenosis, intra-cardiac shunts (eg, ventricular septal defect), or direct trauma (eg, catheters) exist. The turbulence of intracardiac blood flow that results from such abnormalities can lead to further irregularities of the endothelial surfaces that facilitate platelet and fibrin deposi-tion. These factors produce a potential nidus for colonization and infection.
2. Transient bacteremia is common, but it is usually of no clinical importance. Often seen for a few minutes after a variety of dental procedures, it has also been shown to develop after normal childbirth and manipulations such as bronchoscopy, sigmoi-doscopy, cystoscopy, and some surgical procedures. Even simple activities such as tooth brushing or chewing candy can cause such bacteremia. The organisms responsi-ble for transient bacteremia are the common surface flora of the manipulated site such as viridans streptococci (oropharynx) and are usually of low virulence. Other, more virulent strains may also be involved, however; for example, intravenous drug abuse may lead to transient bacteremia with Staphylococcus aureus or a variety of Gram-negative aerobic and anaerobic bacteria. Whether or not the organisms causing bac-teremia (or fungemia) are of high virulence, they can colonize and multiply in the heart if local endothelial changes are suitable.
3. Circulating organisms adhere to the damaged surface, followed by complement acti-vation, inflammation, fibrin, and platelet deposition and further endothelial damage at the site of colonization. The resulting entrapment of organisms in the thrombotic “mesh” of platelets, fibrin, and inflammatory cells leads to a mature vegetation, which protects the organisms from host humoral and phagocytic immune defenses, and to some extent from antimicrobial agents. As a result, the infection can be exceedingly difficult to treat. The vegetation can also create greater hemodynamic alterations in terms of obstruction to flow and increased turbulence. Parts of vegetations may break off and be deposited in smaller blood vessels (embolization) with resultant obstruc-tion and secondary sites of infection. Emboli may be transported to the brain or coro-nary arteries, for example, with disastrous results.
Another phenomenon shown to contribute to the infective endocarditis syndrome is the development of circulating immune complexes of microbial antigen and antibody. These complexes can activate complement and contribute to many of the peripheral mani-festations of the disease, including nephritis, arthritis, and cutaneous vascular lesions.
Frequently, there is a widespread stimulus to host cellular and humoral immunity, particularly if the infection continues for more than about 2 weeks. This condition is char-acterized by hyperglobulinemia, splenomegaly, and the occasional appearance of macrophages in the peripheral blood. Some patients develop circulating rheumatoid fac-tor (IgM anti-IgG antibody), which may play a deleterious role by blocking IgG opsonic activity and causing microvascular damage. Antinuclear antibodies, which also appear occasionally, may contribute to the pathogenesis of the fever, arthralgia, and myalgia that is often seen.
In summary, infective endocarditis involves an initial complex of endothelial damage or abnormality, which facilitates colonization by organisms that may be circulating through the heart. This colonization, in turn, leads to the propagation of a vegetation, with its attendant local and systemic inflammatory, embolic, and immunologic complications.
Clinical Features
Infective endocarditis has often been classified by the progression of the untreated dis-ease. Acute endocarditis is generally fulminant with high fever and toxicity, and death may occur in a few days or weeks. Subacute endocarditis progresses to death over weeks to months with low-grade fever, night sweats, weight loss, and vague constitutional complaints. The clinical course is substantially related to the virulence of the infecting or-ganism; S. aureus, for example, usually produces acute disease, whereas infections by the otherwise avirulent viridans streptococci are more likely to be subacute. Before the ad-vent of antimicrobial therapy, death was considered inevitable in all cases. Physical find-ings often include a new or changing heart murmur, splenomegaly, various skin lesions (petechiae, splinter hemorrhages, Osler’s nodes, Janeway’s lesions), and retinal lesions.
Complications include the risk of congestive heart failure as a result of hemodynamic alterations, rupture of the chordae tendinea of the valves, or perforation of a valve. Abscesses of the myocardium or valve ring can also develop. Other complications relate to the immunologic and embolic phenomena that can occur. The kidney is commonly affected, and hematuria is a typical finding. Renal failure, presumably from immune com-plex glomerulonephritis, is possible. Left-sided endocarditis can readily lead to coronary artery embolization and “mycotic” aneurysms;. In addition, more distant emboli to the central nervous system can lead to cere-bral infarction and infection. Right-sided endocarditis often causes embolization and infarction or infection in the lung.
Etiologic Agents
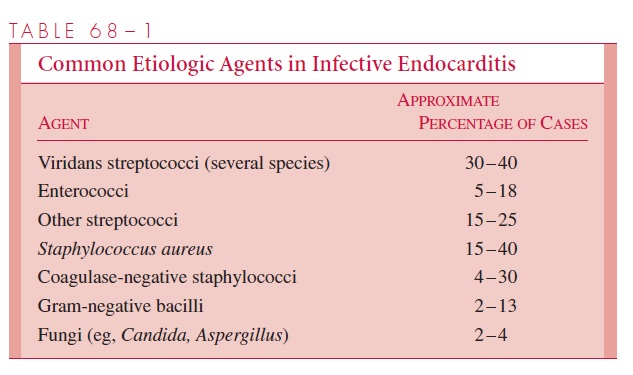
Table 68 – 1 summarizes the most common causes of infective endocarditis. Alpha-hemolytic streptococci and enterococci are involved in just over 50% of the cases. In the so-called culture-negative group, infective endocarditis is diagnosed on clinical grounds, but cultures do not confirm the etiologic agent. This group of patients is difficult to treat, and the overall prognosis is considered poorer than when a specific etiology has been determined. Negative cultures may result from (1) prior antibiotic treatment; (2) fungal endocarditis with entrapment of these relatively large organisms in capillary beds; (3) fastidious, nutritionally deficient, or cell wall – deficient organisms that are difficult to isolate; (4) infection caused by obligate intracellular parasites, such as chlamydiae (Chlamydia psittaci), rickettsiae (Coxiellaburnetii), Rochalimaea species, or viruses; (5) immunologic factors (eg, antibody acting oncirculating organisms); or (6) subacute endocarditis involving the right side of the heart, in which the organisms are filtered out in the pulmonary capillaries.
Some special circumstances alter the relative etiologic possibilities, such as intra-venous drug addiction, prosthetic valves, and immunocompromise. The major associations in these cases are summarized in Table 68 – 2.

General Diagnostic Approaches
The diagnosis of infective endocarditis is usually suspected on clinical grounds; however, the most important diagnostic test for confirmation is the blood culture. In untreated cases, the organisms are generally present continuously in low numbers (1 to 20/mL) in the blood. If an adequate volume of blood is obtained, the first culture will be positive in over 95% of cultur-ally confirmed cases. Most authorities recommend three cultures over 24 hours to ensure detection, and an additional three if the first set is negative. Multiple cultures yielding the same organism support the probability of an intravascular or intracardiac infection. In acute endocarditis, the urgency of early treatment may require collection of only two or three cultures within a few minutes so that antimicrobial therapy can begin.
Cardiologic procedures such as transthoracic or transesophageal echocardiography can delineate the nature and size of the vegetations and progression of disease. They are also helpful in prediction of some complications such as embolization.
General Principles of Management
Because of the nature of the lesions and their pathogenesis, response to therapy may be slow and cure is sometimes difficult. Therefore, specific antimicrobial therapy must be aggressive, using agents that are bactericidal (rather than bacteriostatic) and can be given in amounts that achieve high continuous blood levels without causing toxicity to the pa-tient. Treatment may involve a single antimicrobial if the organism is highly susceptible in vitro, or antimicrobial combinations if synergistic effects are possible (eg, a penicillin and an aminoglycoside for enterococcal endocarditis). Parenteral therapy is begun to pro-duce adequate blood levels, and the patient may need to be monitored frequently to en-sure antimicrobial activity in the serum sufficient to kill the organisms without causing unnecessary toxicity. Therapy is usually prolonged, lasting longer than 4 weeks in most cases. In some cases, surgery may be required to excise the diseased valve and replace it with a valvular prosthesis. The decision for surgery is sometimes difficult, requiring con-sultation with both a cardiologist and a surgeon.
Prophylaxis can prevent the development of endocarditis in persons with known con-genital or acquired cardiac lesions that predispose to bacterial endocarditis. When they un-dergo procedures known to cause transient bacteremia (eg, dental manipulations or surgical procedures involving the upper respiratory, gastrointestinal, or genitourinary tracts), administration of high doses of antimicrobics is begun just before the procedure and continued for 6 to 12 hours thereafter. An example of prophylaxis is the case of a pa-tient with rheumatic valvular disease who is planning to undergo dental work. The organ-ism most likely to produce transient bacteremia would be a penicillin-sensitive member of the oral flora, especially viridans streptococci. Thus, an intramuscular dose of penicillin or ampicillin within 30 minutes before the procedure, followed by a high dose of intramuscu-lar penicillin or oral amoxicillin 6 hours later, would be expected to afford protection. Several regimens similar to this approach are recommended, depending on the patient, the nature of the procedure, and the organisms that might be expected to be involved.
Mycotic Aneurysm
The term mycotic aneurysm is somewhat misleading, because it suggests infection by fungi. Originally used by Sir William Osler to describe the mushroom-shaped arterial aneurysm that can develop in patients with infective endocarditis, the term now applies to infection with any organism that causes inflammatory damage and weakening of an arter-ial wall with subsequent aneurysmal dilatation. This sequence can progress to rupture, with a fatal outcome.
Arterial infection can result from direct extension of an intracardiac infection or from septic microemboli from a cardiac focus, with seeding of vasa vasorum within the arterial wall. In addition to infective endocarditis, other predisposing factors include damaged ar-terial intima by atherosclerotic plaques, vascular thrombi, congenital malformations, trauma, or spread from a contiguous focus of infection directly into the artery. The clini-cal features vary according to the site of involvement. Common findings may include pain at the site of primary arterial supply (eg, back or abdominal pain in abdominal aortic infections) and fever. In many cases, the initial presentation is the result of a catastrophic hemorrhage, particularly intracerebral aneurysms. The etiologic agents, diagnosis, and management are similar to infective endocarditis.
Suppurative Thrombophlebitis
Suppurative (or septic) thrombophlebitis is an inflammation of a vein wall frequentlyassociated with thrombosis and bacteremia. There are four basic forms: superficial, pelvic, intracranial venous sinus, and portal vein infection (pylephlebitis). With the steadily increasing use of intravenous catheters, the incidence of superficial throm-bophlebitis has risen and represents a major complication in hospitalized patients.
The pathogenesis involves thrombus formation, which may result from trauma to the vein, extrinsic inflammation, hypercoagulable states, stasis of blood flow, or combinations of these factors. The thrombosed site is then seeded with organisms, and a focus of infec-tion is established. In superficial thrombophlebitis, an intravenous cannula or catheter may cause local venous wall trauma, as well as serve as a foreign body nidus for throm-bus formation. Infection develops if bacteria are introduced by intravenous fluid, local wound contamination, or bacteremic seeding from a remote infected site.
Thrombophlebitis of pelvic, portal, or intracranial venous systems most often occurs as a result of direct extension of an infectious process from adjacent structures, or from venous and lymphatic pathways near sites of infection. For example, infections of in-tracranial venous sinuses usually result from orbital or sinus infections (causing cav-ernous sinus thrombophlebitis) or from infections of the mastoid and middle ear (causing lateral and sagittal sinus thrombophlebitis). Pelvic thrombophlebitis is a potential result of intrauterine infection (endometritis), particularly after pelvic surgery or 2 to 3 weeks after childbirth. Pelvic or intra-abdominal infections may also spread to the portal venous system to produce pylephlebitis.
Clinical Features
Common features often include fever and inflammation over the infected vein. Pelvic or portal vein thrombophlebitis is usually associated with high fever, chills, nausea, vomiting, and abdominal pain. Jaundice may develop in portal vein infections. Intracranial throm-bophlebitis varies in its presentation. Headache, facial or orbital edema, and neurologic deficits are variably present; for example, cavernous sinus thrombophlebitis often causes palsies of the third through sixth cranial nerves. Complications include extension of suppu-rative infection into adjacent structures, further propagation of thrombi, bacteremia, and septic embolization. Embolization from pelvic or leg veins is to the lungs and pulmonary embolism with infarction may be the presenting manifestation of the remote infection.
Etiologic Agents
The major infectious causes of suppurative thrombophlebitis are outlined in Table 68 – 3. In superficial thrombophlebitis, which often follows intravenous therapy, organisms that are common nosocomial offenders predominate (S. aureus, Gram-negative aerobes). Deeper infections are more frequently caused by organisms that reside on adjacent mu-cous membranes (eg, Bacteroides species in intestinal and vaginal sites) or commonly in-fect adjacent sites (eg, Haemophilus influenzae and S. pneumoniaein acute otitis media and sinusitis).

General Diagnostic Approaches
The diagnosis is often suspected on clinical grounds and from associated events known to create predisposition to such infections (eg, surgery, presence of indwelling venous can-nulas). Direct cultures of the infected site or blood cultures usually yield the infecting or-ganism, because bacteremia is often present. Radiologic procedures, including scanning methods, may be necessary to localize the process and support the diagnosis. In some cases, surgical exploration is required, both for definitive treatment and to obtain speci-mens for cultures.
General Principles of Management
The choice of antimicrobial agents is based on culture and susceptibility test results, or in
the absence of microbiologic data, the most likely possibilities listed in Table 68–3. Other important aspects of management include prompt removal of possible offending sources, such as intravenous catheters, vigorous treatment of adjacent infections, and sometimes surgical excision and drainage. Severe cases may also benefit from systemic anticoagulant therapy to prevent further propagation of thrombi and embolization.
Many cases are preventable. Unnecessary, long-term intravenous cannulation should be avoided. Whenever possible, it is better to use short needles such as “scalp vein cannulas than venous catheters or plastic cannulas. Careful asepsis is essential with all intravenous procedures to prevent contamination of intravenous fluids, tubing, and the site of venous entry.
Intravenous Catheter Bacteremia
A variant of intravascular infection develops when a medical device such as an intra-venous catheter or any of several types of monitoring devices placed in the bloodstream becomes colonized with microorganisms. The event itself does not have immediate clini-cal significance but, unlike transient bacteremia from manipulation of normal floral sites, the bacteremia continues. This persistence greatly increases the chances of secondary complications such as infective endocarditis and metastatic infection, depending on any underlying disease and the virulence of the organism involved.
The organisms involved are usually those found in the skin flora, such as S. epider-midis, Corynebacterium jeikeium, or S. aureus. In debilitated patients already onantimicrobial therapy, Candida species may be involved. Occasionally, the sources of con-tamination are the intravenous solutions themselves rather than the skin. In these cases, members of the Enterobacteriaceae, Pseudomonas, or other Gram-negative rods are more likely.
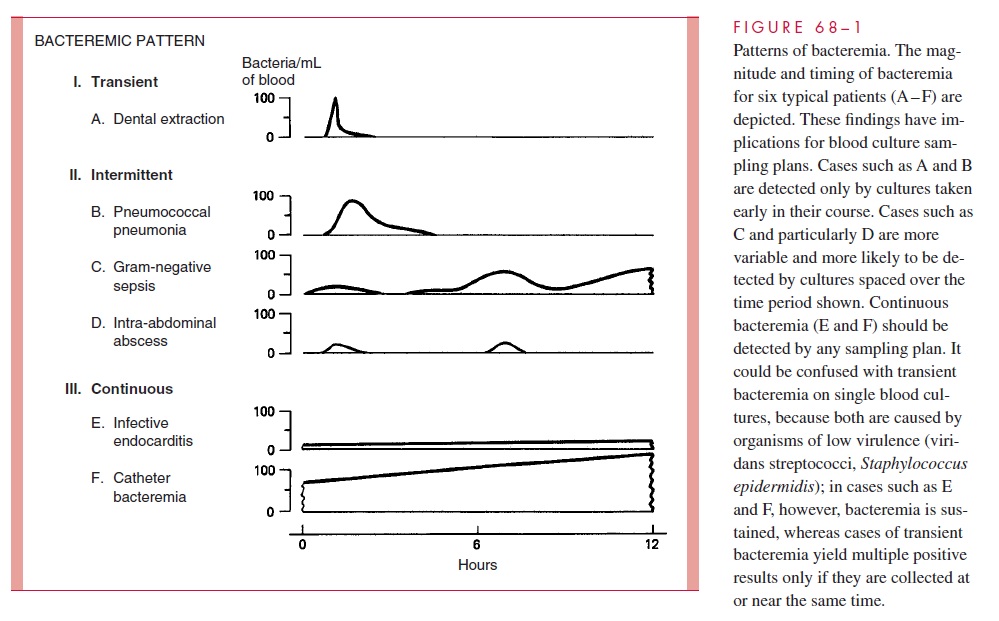
The clinical findings in catheter bacteremia are usually mild despite large numbers of organisms in the bloodstream (Fig 68 – 1). In addition to low-grade fever, signs of inflam-mation may or may not be present. Management includes removal of the contaminated catheter. Antimicrobial therapy alone often does not eradicate the organisms in the pres-ence of a foreign body (the catheter).
Related Topics