Chapter: Medical Microbiology: An Introduction to Infectious Diseases: Intravascular Infections, Bacteremia, and Endotoxemia
Blood Culture
BLOOD CULTURE
The primary means for establishing a diagnosis of sepsis is by blood culture. The micro-biologic principles involved are the same as with any culture. A sample of the patient’s blood is obtained by aseptic venipuncture and cultured in an enriched broth or, after spe-cial processing, on plates. Growth is detected, and the organisms are isolated, identified, and tested for antimicrobial susceptibility. Because of the importance of blood cultures in the diagnosis and therapy of most bacterial and fungal infections, considerable attention must be paid to details of sampling if the prospects of obtaining a positive culture are to be maximized. The approach to blood culture must be tailored to the individual patient; no single procedure is best for all individuals. The important features are described below.
Blood Culture Sampling
Venipuncture
Before venipuncture, the skin over the vein must be carefully disinfected to reduce the probability of contamination of the blood sample with skin bacteria. Although it is not possible to “sterilize” the skin, quantitative counts can be markedly reduced with a com-bination of 70% alcohol and an iodine-based antiseptic. Mechanical cleansing is as im-portant as use of the antiseptic. Poor phlebotomy technique such as repalpating the vein after the preparation is related to introduction of contaminants. Blood is ideally drawn di-rectly into a blood culture bottle or a sterile blood collection vacuum tube containing an anticoagulant free of antimicrobial properties. Sodium polyanethol sulfonate is currently preferred; other anticoagulants such as citrate and ethylenediaminetetraacetic acid have antibacterial activity. Blood should not be drawn through indwelling venous or arterial catheters unless it cannot be obtained by venipuncture.
Volume
The number of organisms present in blood is often low ( < 1 organism/mL) and cannot be predicted in advance. Thus, small samples yield fewer positive cultures than larger ones. For example, as the volume sampled increases from 2 to 20 mL, the diagnostic yield in-creases by 30 to 50%. Samples of at least 10 mL should be collected from adult patients. The same principles apply with infants and young children, but the sample size must be reduced to take account of the smaller total blood volume of a child. Although it should be possible to obtain at least 1 mL, smaller volumes should still be cultured because bac-teremia at levels of more than 1000 bacteria/mL is found in some infants.
Number
If the volume is adequate, it is rarely necessary to collect more than two or three blood cultures to achieve a positive result. In intravascular infections (eg, infective endocardi-tis), a single blood culture is positive in more than 95% of cases. Studies of sequential blood cultures from bacteremic patients without endocarditis have yielded 80 to 90% pos-itive results on the first culture, more than 90 to 95% with two cultures, and 99% in at least one of a series of three cultures.
Timing
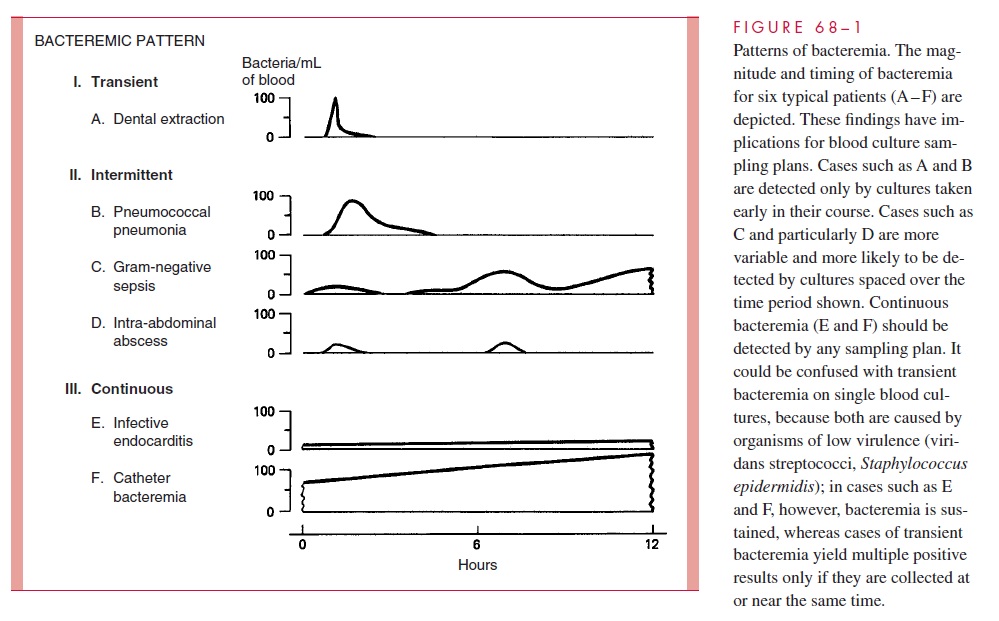
The best timing schedule for a series of two or three blood cultures is dependent on the bacteremic pattern of the underlying infection and the clinical urgency of initiating an-timicrobial therapy. Figure 68 – 1 illustrates some typical bacteremic patterns that can be related to the probability of obtaining positive blood cultures. Transient bacteremia is usually not detected, because organisms are cleared before the appearance of any clinical findings suggesting sepsis. The continuous bacteremia of infective endocarditis is usually readily detected, and timing is not critical. Intermittent bacteremia presents the greatest challenge because fever spikes generally occur after, rather than during, the bacteremia. Little is known about the periodicity of bloodstream invasion, except that the bacteremia is more likely to be present and sustained in the early acute stages of infection. Closely spaced samples are less likely to detect the organism than those spaced an hour or more apart. In urgent situations, when antimicrobial therapy must be initiated, two or three samples should be collected at brief intervals and therapy begun as soon as possible. It is generally not useful to collect blood cultures while the patient is receiving antimicrobics unless none were collected before therapy or there is a change in the clinical course sug-gesting superinfection. The laboratory should be advised when such cultures are submit-ted, because it is sometimes possible to inactivate an antimicrobic, for example, with beta-lactamases.
Laboratory Processing
The basic blood culture procedure of incubating blood in an enriched broth is quite sim-ple, but considerable effort must be expended to ensure detection of the broadest range of organisms in the least possible time. Daily examination of cultures for 1 week or more and a routine schedule of stains and/or subcultures of apparently negative cultures are re-quired to detect organisms such as H. influenzae or N. meningitidis, which usually do not produce visual changes in the broth. Direct plating of blood onto blood or chocolate agar is accomplished in a system that concentrates the blood by centrifugation following lysis of the erythrocytes. This is particularly useful for bacterial quantification and rapid identi-fication. Automated blood culture systems detect metabolic activity (primarily CO2 gener-ation) in broth culture for initial detection in place of the conventional visual and staining examinations. These systems detect growth sooner than conventional methods but still require subculture for confirmation, identification, and susceptibility testing.
Isolation of fungi is favored by ensuring maximum aerobic conditions in direct plat-ing systems and broth bottles. Conversely, anaerobes are recovered best when a highly re-duced environment is provided for plates and broths. Some bacteria, such as Leptospira, are not isolated by routine blood culture procedures. The laboratory must be notified in advance so special media can be used.
Because the blood is normally sterile, the interpretation of blood cultures growing a pathogenic organism is seldom a problem. The major problem is the differentiation of agents causing transient bacteremia and skin contamination from those opportunists asso-ciated with an intravascular or extravascular infection. Transient bacteremia is of short duration (see Fig 68 – 1), is associated with manipulation of or trauma to a site possessing a normal flora, and involves species indigenous to that site. Despite skin disinfection, 2 to 4% of venipunctures result in contamination of the culture with small numbers of cuta-neous flora such as S. epidermidis, corynebacteria (diphtheroids), and propionibacteria. The presence of these organisms in blood cultures can be considered a result of skin con-tamination unless quantitative procedures indicate large numbers ( 5 organisms/mL) or repeated cultures are positive for the same organism. These findings should suggest diseases such as infective endocarditis or catheter bacteremia.
Related Topics