Chapter: Modern Pharmacology with Clinical Applications: Histamine and Histamine Antagonists
Histamine
HISTAMINE
Sinus problems, hay fever,
bronchial asthma, hives, eczema, contact dermatitis, food allergies, and
reactions to drugs are all allergic reactions associated with the re-lease of
histamine and other autocoids, such as sero-tonin, leukotrienes, and
prostaglandins. Histamine re-lease is frequently associated with various
inflammatory states and may be increased in urticarial reactions,
mas-tocytosis, and basophilia. Histamine also acts as a neu-rotransmitter in
the central nervous system (CNS). Upon release from its storage sites,
histamine exerts ef-fects ranging from mild irritation and itching to
ana-phylactic shock and eventual death.
Histamine is found in animal
tissues and venoms and in many bacteria and plants. Within the human body, the
largest histamine concentrations are in the skin, lungs, and gastrointestinal
mucosa, while concentrations are smaller in almost all other organs and
tissues. Histamine is present in human plasma at relatively low concentra-tions
(usually less than 0.5 ng/mL); in contrast, whole-blood levels can be as high
as 30-fold greater. Substantial quantities of histamine are present in urine,
with excre-tion rates varying from 10 to 40 g per 24 hours.
Synthesis and Storage
Virtually all of the
histamine found in individual organs and tissues is synthesized locally and
stored in subcellu-lar secretory granules. Within
the tissues, the mast cells are the
principal sites of storage; in the blood, the ba- sophils serve this function. Histamine
is also present in neurons of the
CNS, where it acts as a neurotransmitter.
Histamine is synthesized from
the amino acid histi-dine by an action of the enzyme histidine decarboxylase (Fig. 38.1). Following synthesis, histamine
is either rap-idly inactivated or stored in the secretory granules of mast
cells and basophils as an inactive complex with proteases and heparin sulfate
or chondroitin sulfate.
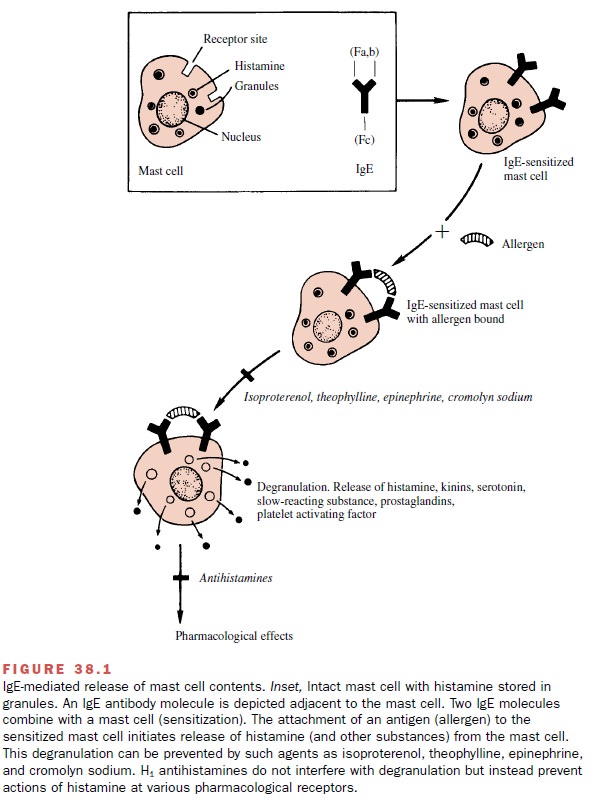
Release from Storage Sites
Histamine can be released
from mast cell granules in two ways, both of which have pharmacological impor- tance.
Endogenous or exogenous compounds can pro-mote an exocytotic release of
histamine without cell de-struction or lysis. Alternatively, histamine can be
re-leased from mast cells by a variety of nonexocytotic processes, including
mast cell lysis, modification of mast cell membranes, and physical displacement
of histamine.
Both exocytotic and nonexocytotic mechanisms can contribute to adverse drug reactions that involve hista-mine release. Histamine is only one of several potent physiological mediators that are released from mast cells; the other substances can also contribute to the overall immediate hypersensitivity reaction.
Antigen-Mediated Histamine Release
Specific antigen–antibody
interactions initiate the degran-ulation of tissue mast cells and blood
basophils as part of the immediate hypersensitivity reaction. Immunoglobulin E
(IgE) antibodies (reaginic antibodies) directed against an allergenic substance
attach to the outer surface of the cell membrane and initiate a series of
biochemical events that culminate in the release of the secretory granule
con-tents (Fig. 38.1).Although allergens are the most frequent initiators of
immediate hypersensitivity reactions, certain drugs, particularly in
association with endogenous high-molecular-weight molecules, may also promote
the sensi-tization process and mast cell degranulation on subse-quent drug
exposure.
Certain endogenous and
exogenous compounds modulate the antigen-mediated release of histamine from
sensitized tissues. Histamine inhibits its own re-lease in skin mast cells and
blood basophils by binding to H2 histamine receptors, which when
activated, inhibit degranulation. This feedback inhibition does not appear to
occur in lung mast cells. Agonists of β2-adrenoceptors inhibit antigen-induced
histamine release from mast cells, whereas muscarinic and α-adrenergic agonists en-hance
mast cell degranulation.
Non–Antigen-Mediated Release of Histamine
Histamine may be released
from mast cells by mecha-nisms that do not require prior sensitization of the
im-mune system. Drugs, high-molecular-weight proteins, venoms, and other
substances that damage or disrupt cell membranes can induce the release of
histamine. Any thermal or mechanical stress of sufficient intensity also will
result in histamine release. Cytotoxic com-pounds, may release histamine as the
result of disrup-tion of cell membranes.
Drugs, particularly organic
bases, may release hista-mine from mast cells by physically displacing the
amine from its storage sites. Morphine, codeine, d-tubocu-rarine, guanethidine, and radiocontrast media can re-lease
histamine from mast cells. Basic polypeptides, such as bradykinin, neurotensin,
substance P, somatostatin, polymyxin B, and the anaphylatoxins resulting from
complement activation, also stimulate histamine release. Venoms often contain
basic polypeptides as well as the histamine-releasing enzyme phospholipase A.
Inactivation of Released Histamine
The inactivation of histamine
is achieved both by enzy-matic metabolism of the amine and by transport
processes that reduce the concentration of the com-pound in the region of its
receptors. Histamine metabo-lism occurs primarily through two pathways (Fig.
38.1). The most important of these involves histamine
N-methyltransferase, which catalyzes the transfer of amethyl group from
S-adenosyl-1-methionine to one of the imidazole nitrogen substitutions, forming
1-methyl-histamine. This enzyme is present in tissues but not in blood.
1-Methylhistamine is converted by monoamine oxidase (MAO) to
1-methylimidazoleacetic acid.
An alternative pathway of
histamine metabolism in-volves oxidative deamination by the enzyme diamine oxidase (histaminase) to
form 5-imidazoleacetic acid. Diamine
oxidase is present in both tissues and blood and plays a particular role in
metabolizing the large concentrations of histamine that may be present in food.
An additional metabolite, N-acetyl
histamine (a conjugate of acetic acid and histamine), can be pro-duced if
histamine is ingested orally. This product may result from metabolism of
histamine by gastrointestinal tract bacteria. Because of its rapid breakdown
after oral administration, histamine produces few systemic effects when given
by this route.
Physiological Effects of Histamine
Histamine mediates a diverse
group of processes rang-ing from vasodilation to gastric acid secretion. It
pro-duces its effects by binding to and activating receptors on the surface of
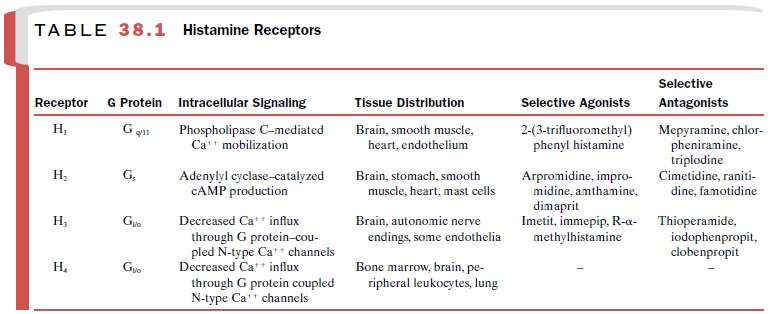
cardiac, smooth muscle, endothelial, neuronal, and other cells. There are at
least four recep-tor populations, H1, H2, H3,
and H4. All four receptor subtypes have been cloned and belong to
the G pro-tein–coupled receptor superfamily. The histamine re-ceptors can be
distinguished on the basis of their post–receptor signal transduction
mechanisms, tissue distribution, and sensitivities to various agonists and
an-tagonists (Table 38.1). Currently, only the H1- and H2-receptors
are targets of clinical drug therapy.
Cardiovascular System
A slow intravenous injection
of histamine produces marked vasodilation of the arterioles, capillaries, and
venules. This causes a fall in blood pressure whose mag-nitude depends on the
concentration of histamine in-jected, the degree of baroreceptor reflex
compensation, and the extent of histamine-induced release of adrenal
catecholamines. Vasodilation of cutaneous blood vessels reddens the skin of the
face, while a throbbing headache can result from vasodilation of brain
arterioles. Vasodilation is mediated through both H1- and H2-receptors
on vascular smooth muscle. Stimulation of H1-receptors produces a
rapid and short-lived response, whereas stimulation of H2-receptors
produces a more sustained response that is slower in onset. Stimulation of H3-receptors
on sympathetic nerve terminals inhibits the release of norepinephrine and its
associated vaso-constriction.
Histamine increases the
permeability of capillaries and postcapillary vessels, resulting in passage of
fluid and protein into the extracellular space and eventually edema. This H1-receptor–mediated
process is responsi-ble for the urticarial effect of histamine on the skin
(hives).
In addition to its effects on
the vasculature, hista-mine exerts direct positive inotropic and chronotropic
effects on the heart through the stimulation of H2-receptors. H3-receptors
on sympathetic nerve terminals in the heart decrease norepinephrine release;
however, this effect appears to be significant only during stress states such
as ischemia.
Extravascular Smooth Muscle
Histamine stimulates
bronchiolar smooth muscle con-traction through activation of H1-receptors.
A much smaller bronchodilatory response is evoked by stimula-tion of H2-receptors.
Asthmatics are generally more sensitive to the bronchoconstrictor actions of
histamine than are nonasthmatics.
Histamine is able to cause
uterine contraction. Although the magnitude of this effect in humans is
nor-mally small, the large amounts of histamine released during anaphylactic
reactions can initiate abortion in pregnant women. Histamine can also stimulate
contrac-tion of gastrointestinal smooth muscle, with large doses able to
produce diarrhea.
Glandular Tissue
Histamine stimulates the secretion of gastric acid and pepsin through
an effect on the H2-receptors of the pari-etal cells of the gastric
mucosa. Secretion of acid is a complex
process that is stimulated by histamine, acetyl-choline, and gastrin and
inhibited by somatostatin. The ability of H2-receptor antagonists to
inhibit the en-hanced gastric acid secretion caused by acetylcholine and
gastrin suggests that histamine release is of primary importance in this
process. Histamine also stimulates secretion by the salivary glands and glands
in the small and large intestines. High concentrations of histamine promote the
release of catecholamines from the adrenal gland.
Nervous System
Postsynaptic H1-
and H2-receptors are responsible for a variety of processes in the
CNS. H1-receptors mediate the maintenance of wakeful states, while H1-
and H2-receptors participate in the regulation of blood pres-sure,
body temperature, fluid homeostasis, and pain sen-sation. Presynaptic H3-receptors
serve as feedback in-hibitors of the release of histamine, norepinephrine, and
other neurotransmitters.
In the periphery, H1-receptors
on sensory neurons in the epidermis and dermis mediate itch and pain,
respec-tively. Autonomic afferent nerve endings may be simi-larly stimulated by
histamine. As in the CNS, presynap-tic H3-receptors act in a feedback
inhibitory capacity.
Lewis Triple Response
The Lewis triple response illustrates the effects of hista-mine on
vascular smooth muscle, vascular endothelium, and sensory nerve endings.
Intradermal injection of as little as 10 g histamine produces three distinct
effects:
·
Dilation of capillaries in the immediate vicin-ity of the injection
results in a local red or blue region (flush).
·
Dilation of arterioles results in an irregular red flare over an area that is generally
wider than that due to the capillary
dilation. The flare probably results from an axon reflex in which histamine
stimulates autonomic nerve endings, causing release of vasodilatory mediators.
·
Swelling (wheal) appears
in the area of capil-lary dilation. The increased permeability of the blood
vessels in this region is responsible for the edema.
In addition to the flush,
wheal, and flare, transient pain and itching result from the effects of
histamine on sensory nerve endings. In sensitized individuals, intra-dermal
injection of specific antigens produces a wheal; this reaction is the basis for
a skin test to quantify the extent of the allergic response.
Anaphylaxis
During an anaphylactic
reaction, large quantities of in-flammatory mediators are rapidly released. The
resul-tant reaction is severe and may threaten the life of the individual. The
introduction of a specific antigen— usually in food or in injected
material—into a sensitized individual can cause the rapid release of mast cell
con-tents, producing a decrease in blood pressure, impaired respiratory
function, abdominal cramps, and urticaria. Extreme and severe anaphylaxis is
life threatening and requires prompt medical intervention.
Clinical Uses of Histamine
Histamine has only minor uses
in clinical medicine. In the past it was used to diagnose pernicious anemia, in
which histamine fails to evoke the usual secretion of gastric acid. Histamine
has been used to assess bronchial hyperreactivity, although this test may be
quite hazardous for asthmatics. Today the main clinical use of histamine is as
a positive control injection for al-lergy skin testing.
Related Topics