Chapter: Mechanical and Electrical : Thermal Engineering : Gas Power Cycles
Gas Power Cycles
GAS POWER CYCLES
PREREQUISITE DISCUSSIONS
Discussion of this gas power
cycles will involve the study of those heat engines in which the working fluid
remains in the gaseous state throughout the cycle. We often study the ideal
cycle in which internal irreversibilities and complexities (the actual intake
of air and fuel, the actual combustion process, and the exhaust of products of
combustion among others) are removed. We will be concerned with how the major
parameters of the cycle affect the performance of heat engines. The performance
is often measured in terms of the cycle efficiency.
INTRODUCTION
Ø The cycle
is defined as the repeated series of operation or processes performed on a
system, so that the system attains its original state.
Ø The cycle
which uses air as the working fluid is known as Gas power cycles.
Ø In the
gas power cycles, air in the cylinder may be subjected to a series of
operations which causes the air to attain to its original position.
Ø The
source of heat supply and the sink for heat rejection are assumed to be
external to the air.
Ø The cycle
can be represented usually on p-V and T-S diagrams.
POWER CYCLES
Ø Ideal
Cycles, Internal Combustion
Ø Otto
cycle, spark ignition
Ø Diesel
cycle, compression ignition
Ø Sterling
& Ericsson cycles
Ø Brayton
cycles
Ø Jet-propulsion
cycle
Ø Ideal
Cycles, External Combustion
Ø Rankine
cycle
IDEAL CYCLES
Ø Idealizations
& Simplifications
Ø Cycle
does not involve any friction
Ø All
expansion and compression processes are quasi-equilibrium processes
Ø Pipes
connecting components have no heat loss
Ø Neglecting
changes in kinetic and potential energy (except in nozzles & diffusers)
GAS POWER CYCLES
Ø Working
fluid remains a gas for the entire cycle
Ø Examples:
Ø Spark-ignition
engines
Ø Diesel
engines
Ø Gas
turbines
Air-Standard Assumptions
Ø Air is
the working fluid, circulated in a closed loop, is an ideal gas
Ø All
cycles, processes are internally reversible
Ø Combustion
process replaced by heat-addition from external source
Ø Exhaust
is replaced by heat rejection process which restores working fluid to initial
state
ENGINE TERMS
Ø Top dead
center
Ø Bottom
dead center
Ø Bore
Ø Stroke
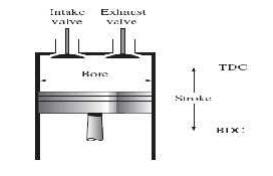
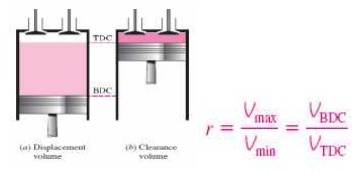
Ø Clearance
volume
Ø Displacement
volume
Ø Compression
ratio
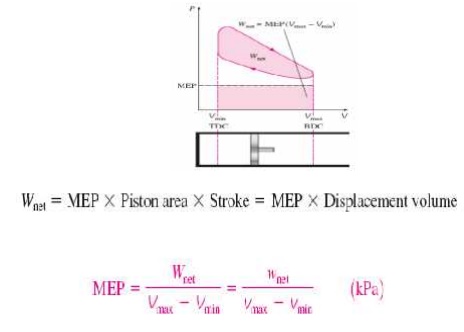
Ø Mean effective pressure (MEP)
CYCLES
AND THEIR CONCEPTS
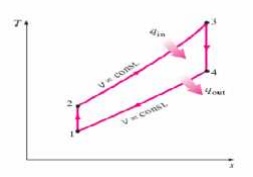
OTTO CYCLE
An Otto cycle is an
idealized thermodynamic cycle that describes the functioning of a typical spark
ignition piston engine. It is the thermodynamic cycle most commonly found in
automobile engines. The idealized diagrams of a four-stroke Otto cycle Both diagrams
Ø Petrol
and gas engines are operated on this cycle
Ø Two
reversible isentropic or adiabatic processes
Ø Two constant volume process
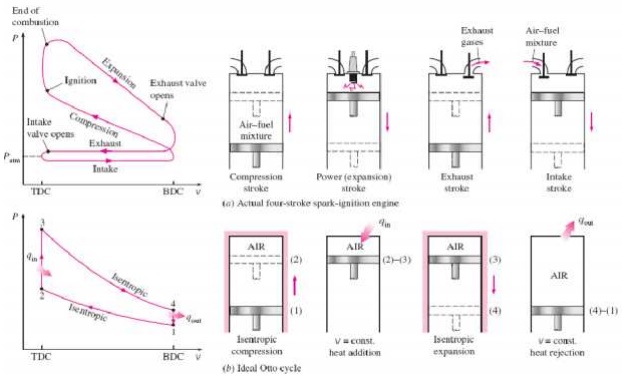
PROCESS OF OTTO CYCLE
Ø Ideal Otto
Cycle
Ø Four
internally reversible processes
o 1-2 Isentropic compression
o 2-3 Constant-volume heat
addition
o 3-4 Isentropic expansion
o 4-1 Constant-volume heat rejection
Thermal efficiency of ideal Otto
cycle:
Since V2= V3
and V4 = V 1
Diesel cycle
The Diesel cycle is a co mbustion
process of a reciprocating internal co mbustion engine. In it, fuel is ignited
by hea t generated during the compression of air in the combustion chamber,
into which fuel is t hen injected.
It is assumed to have constant
pressure during the initial part of the "combustion" phase The Diesel
engine is a
heat engine: it
converts heat into work.
During the bottom isentropic processes (blue), energy is transferred
into the system in the form of work , Win
but by definition (isentropic) no ene rgy is transferred into or out of the
system in the form of heat. During the constant pressure ( red, isobaric)
process, energy enters the syste m as heat
. During the top isentropic processes (yellow), energy is transferred
out of the sy stem in the form of Wout,
but by definition (isen tropic) no energy is transferred into or out of the
system in the form of heat. During the constan t volume (green,isochoric)
process, some of energy flows out of the system as heat through the right
depressurizing process Qout . The wor k that leaves the system is
equal to the work that enters the system plus the difference between the heat
added to the system and the heat that lea ves the system; in other words, net
gain of wo rk is equal to the difference between the heat adde d to the system
and the heat that leaves the system.
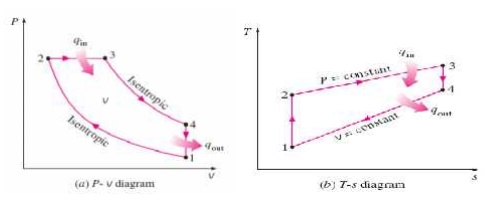
PROCESSES OF DIESEL CYCLE:
Ø 1-2
Isentropic com pression
Ø 2-3
Constant-Pres sure heat addition
Ø 3-4
Isentropic expansion
Ø 4-1
Constant-volume heat rejection
For ideal diesel cycle
Cut off ratio rc
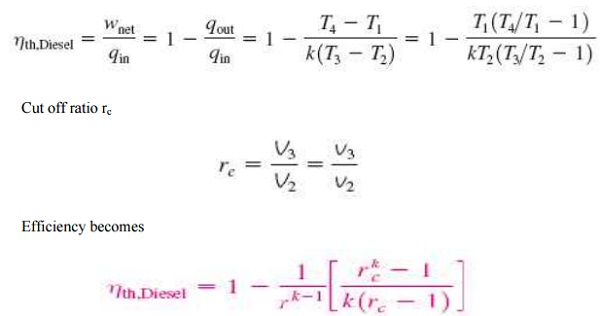
DUAL CYCLE
The dual combustion cycle (also
known as the limited pressure or mixed cycle) is a thermal cycle that is a
combination of the Otto cycle and the Diesel cycle. Heat is added partly at
constant volume and partly at constant pressure, the advantage of which is that
more time is available for the fuel to completely combust. Because of lagging
characteristics of fuel this cycle is invariably used for diesel and hot spot
ignition engines.
Ø Heat addition
takes place at constant volume and constant pressure process .
Ø Combination
of Otto and Diesel cycle.
Ø Mixed
cycle or limited pressure cycle
PROCESS OF DUAL CYCLE

Ø Isentropic
compression
Ø Constant-volume
heat rejection
Ø Constant-pressure
heat addition
Ø Isentropic
expansion
Ø Constant-volume
heat rejection
The cycle is the equivalent air cycle for reciprocating high
speed compression ignition engines. The P-V and T-s diagrams are shown in
Figs.6 and 7. In the cycle, compression and expansion processes are isentropic;
heat addition is partly at constant volume and partly at constant pressure
while heat rejection is at constant volume as in the case of the Otto and
Diesel cycles.
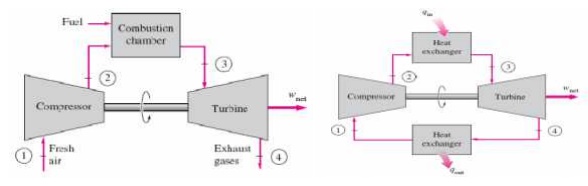
BRAYTON CYCLE
The Brayton cycle is a
thermodynamic cycle that describes the workings of a constant pressure heat
engine. Gas turbine engines and airbreathing jet engines use the Brayton Cycle.
Although the Brayton cycle is usually run as an open system (and indeed must be
run as such if internal combustion is used), it is conventionally assumed for
the purposes of thermodynamic analysis that the exhaust gases are reused in the
intake, enabling analysis as a closed system. The Ericsson cycle is similar to
the Brayton cycle but uses external heat and incorporates the use of a
regenerator.
Ø Gas
turbine cycle
Ø Open vs
closed system model
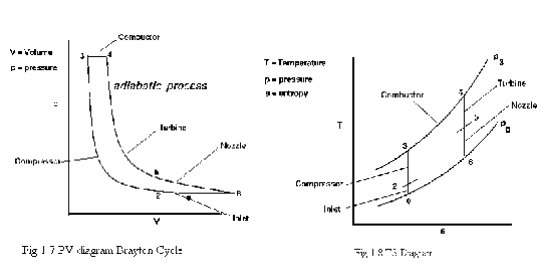
With cold-air-standard assumptions
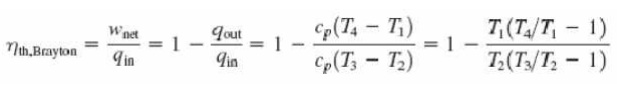
Ø Since
processes 1-2 and 3-4 are isentropic, P2 = P3 and P4 = P1

REAL TIME APPLICATIONS
PETROL ENGINES
Ø Datsun Go
Ø Hyundai
Xcent
Ø Maruti
Suzuki Celerio
Ø Volkswagen
Vento
Ø Nissan
Terrano
DIESEL ENGINES
Ø Isuzu
Diesel Cars
Ø Datsun
Diesel Cars
Ø Ashok
Leyland Diesel Cars
GAS TURBINES
Ø Indraprastha
(Delhi) CCGT Power Station India
Ø Kovilkalappal
(Thirumakotai) Gas CCGT Power Station India
Ø Lanco
Tanjore (Karuppur) CCGT Power Plant India
TECHNICAL TERMS
Ø TDC: Top
Dead Center: Position of the piston where it forms the smallest volume
Ø BDC: Bottom Dead Center: Position of the piston
where it forms the largest volume
Ø Stroke: Distance between TDC and BDC
Ø Bore :
Diameter of the piston (internal diameter of the cylinder)
Ø Clearance
volume: ratio of maximum volume to minimum volume VBDC/VTDC
Ø Engine displacement
: (no
of cylinders) x (stroke length) x (bore area) (usually given in cc or
liters)
Ø MEP: mean
effective pressure: A const. theoretical pressure that if acts on piston produces
work same as that during an actual cycle
Ø Gas Power
Cycles: Working fluid remains in the gaseous state through the cycle.
Sometimes useful to study an idealised cycle in which internal
irreversibilities and complexities are removed. Such cycles are called:Air
Standard Cycles
Ø The mean
effective pressure (MEP): A fictitious pressure that, if it were
applied to the piston during the power stroke, would produce the same
amount of net work as that produced during the actual cycle.
Ø Thermodynamics:
Thermodynamics
is the science of the relations between heat ,work and the properties of
system
Ø Boundary:
System
is a fixed and identifiable collection of matter enclosed by a real or imaginary
surface which is impermeable to matter but which may change its shape or
volume. The surface is called the boundary
Ø Surroundings:
Everything
outside the system which has a direct bearing on the system's behavior.
Ø Extensive
Property: Extensive properties are those whose value is the sum of the
values for each subdivision of the system, eg mass, volume.
Ø Intensive
Property: Properties are those which have a finite value as the size of
the system approaches zero, eg pressure, temperature, etc.
Ø Equilibrium:
A
system is in thermodynamic equilibrium if no tendency towards spontaneous
change exists within the system. Energy transfers across the system
disturb the equilibrium state of the system but may not shift the system
significantly from its equilibrium state if carried out at low rates of change.
I mentioned earlier that to define the properties of a system, they have to be
uniform throughout the system.
Therefore to define the state of system, the
system must be in equilibrium. Inequilibrium of course implies non-uniformity
of one or more properties).
Ø Isentropic
process: Isentropic process is one in which for purposes of engineering
analysis and calculation, one may assume that the process takes place from
initiation to completion without an increase or decrease in the entropy of the
system, i.e., the entropy of the system remains constant.
Ø Isentropic
flow: An isentropic flow is a flow that is both adiabatic and
reversible. That is, no heat is added to the flow, and no energy
transformations occur due to friction or dissipative effects. For an isentropic
flow of a perfect gas, several relations can be derived to define the pressure,
density and temperature along a streamline.
Ø Adiabatic
heating: Adiabatic heating occurs when the pressure of a gas is
increased from work done on it by its surroundings, e.g. a piston.
Diesel engines rely on adiabatic heating during their compression.
Ø Adiabatic
cooling: Adiabatic cooling occurs when the pressure of a substance is
decreased as it does work on its surroundings. Adiabatic cooling occurs in
the Earth's atmosphere with orographic lifting and lee waves, When the pressure
applied on a parcel of air decreases, the air in the parcel is allowed to
expand; as the volume increases, the temperature falls and internal energy
decreases.
SOLVED PROBLEMS
1. In an Otto cycle air at 1bar and 290K is
compressed isentropic ally until the pressure is 15bar The heat is added at
constant volume until the pressure rises to 40bar. Calculate the air standard
efficiency and mean effective pressure for the cycle. Take Cv=0.717 KJ/Kg K and
Runiv = 8.314KJ/Kg K.
Given Data:
Pressure
(P1) = 1bar = 100KN/m2
Temperature(T1)
= 290K
Pressure
(P2) = 15bar = 1500KN/m2
Pressure
(P3) = 40bar = 4000KN/m2
Cv =
0.717 KJ/KgK
Runiv
= 8.314 KJ/Kg K
To Find:
i) Air Standard Efficien cy (ηotto) ii)
Mean Effective Press ure (Pm)
Solution:
Here
it is g Runiv = 8.314 KJ/Kg K
We
know

2.
Estimate the lose in air st andard efficiency for the diesel engine for the c
ompression ratio 14 and the cutoff change s from 6% to 13% of the stroke.

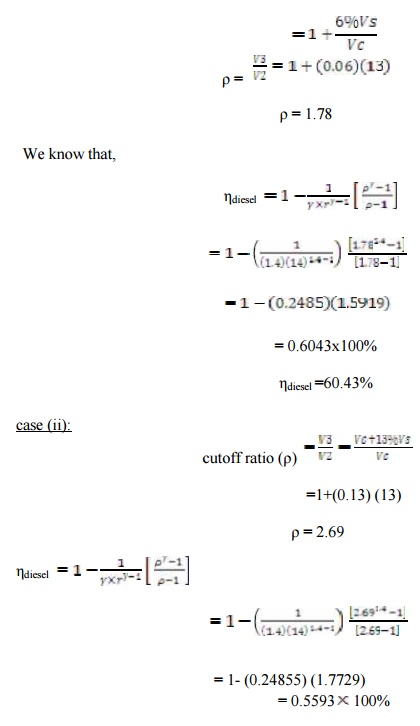
=55.93%
Lose in
air standard efficiency = (ηdiesel
CASE(i) ) - (ηdiesel
CASE(i) )
= 0.6043-0.5593
= 0.0449
= 4.49%
3.
The compression ratio o f an air standard
dual cycle is 12 and the maximum pressure on the cycle is limited to 70bar. Th
e pressure and temperature of the cycle at the b eginning of compression
process are 1bar a nd 300K. Calculate the thermal efficiency and M ean
Effective Pressure. Assume cylinder bore = 250mm, Stroke length = 300mm,
Cp=1.005K J/Kg K, Cv=0.718KJ/Kg K.
Given
data:
Assume Qs1
= Qs2
Compression
ratio (r) = 12
Maximum
pressure (P3) = (P4) = 7000 KN/m2
Temperature
(T1) = 300 K
Diameter
(d) = 0.25m
Stroke
length (l) = 0.3m
To find:
Dual
cycle efficiency (ηdual)
Mean
Effective Pressure (P m)
Solution:
By Process
1-2:
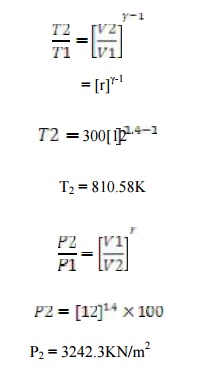


= 832.58/0.0147
Pm = 56535 KN/m2
4.A diesel engine
operating an air standard
diesel cycle has 20cm bore
and 30cmstroke.the clearance volu me is 420cm3.if the fuel is
injected at 5% of the stroke,find the air standard efficiency.
Given Data:-
Bore
diameter (d) =20cm=0.2mk
Stroke,
(l) =30cm=0.3m
Clearance volume, (v2
) =420cm3=420/1003= ![]()
![]()
![]() m3
m3
To Find:-
Air standard efficiency, (diesel)
Solution:-
Compression ratio,
r = v1/v2
= (vc+vs)/vc
We know that,
Stroke vo lume, vs=area*length

Related Topics