Chapter: Modern Analytical Chemistry: Kinetic Methods of Analysis
Flow Injection Analysis: Instrumentation
Instrumentation
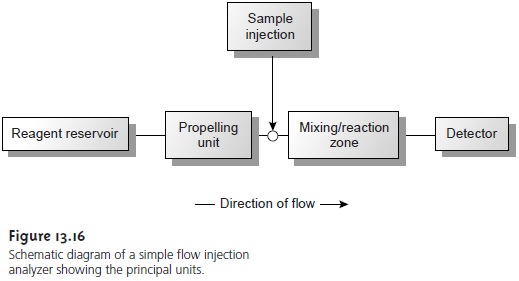
The basic components of a flow injection analyzer
are shown in Figure 13.16
and include a unit
for propelling the
carrier stream, a means for
injecting the sample into the carrier stream,
and a detector for monitoring the composition of the carrier stream. These units are connected by a transport
system that provides
a means for bringing together separate
channels and that enables an appropriate mixing of the sample with the carrier
stream. Separations modules
also may be incorporated in the
flow injection analyzer. Each of these
components is considered in greater detail in this section.
Propelling Unit
The propelling unit
is used to move the
carrier stream in the main channel, as well as any additional reagent streams in secondary channels,
through the flow injection analyzer. Although several different propelling
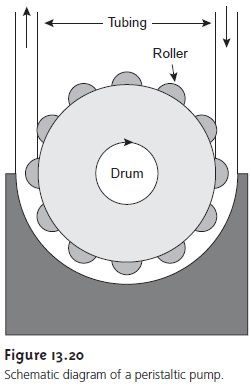
units have been used, the most common is a peristaltic pump. A peristaltic pump consists of a set of
rollers attached to the outside
of a rotating drum (Figure
13.20). Tubing from the
reagent reservoirs is placed in between the rollers and a fixed
plate. As the drum ro- tates the rollers squeeze
the tubing, forcing
the contents of the tubing
to move in the
direction of the rotation. Peristaltic pumps are capable
of providing a constant
flow rate, which is controlled by the drum’s
speed of rotation and the inner
diame- ter of the tubing. Flow rates from 0.0005–40 mL/min
are possible, which
is more than adequate
to meet the needs of FIA, for which flow rates of 0.5–2.5 mL/min
are common. One limitation to peristaltic pumps
is that they
produce a pulsed
flow, particularly at higher
flow rates, which
may lead to oscillations in the signal.
Injector
The
sample, typically 5–200
μL, is placed
in the carrier
stream by injec- tion. Although syringe injections through a rubber septum are used, a more com- mon
means of injection is the rotary,
or loop, injector
used in HPLC.
This type of injector provides reproducible injection
volumes and is easily adaptable
to automation, a feature that is particularly impor- tant when high sampling rates
are desired.
Detector
Detection in FIA may
be accomplished using
many of the
electrochemi- cal and optical detectors used in HPLC. These detectors were discussed and
are not considered further in this
section. In addition, FIA detectors also have been designed
around the use of ion-selective electrodes and atomic ab-
sorption spectroscopy.
Transport System
The heart of a flow injection analyzer
is the transport system
used to bring together the carrier stream,
the sample, and any reagents
that must react with the sample
to generate the desired signal.
Each reagent reservoir con- nected to the flow injection analyzer is considered a separate channel.
All channels must merge
before the carrier
stream reaches the detector, with the merging
points determined by the chemistry involved
in the method. The completed assembly of channels is called a manifold.
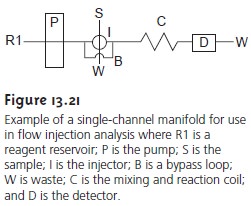
The simplest
manifold includes only a single
channel, the
basic outline of which is shown in Figure 13.21.
This type of manifold is commonly used
for direct analyses that
do not require
a chemical reaction. In this case
the carrier stream
only serves as a means for
transporting the sample
to the detector rapidly and repro- ducibly. For example, this manifold design
has been used as a means of sample in- troduction in atomic absorption spectroscopy, achieving sampling
rates as high asDW700 samples/h. This manifold also is used for determining a sample’s pH or the concentration of metal ions using ion-selective electrodes.
A single-channel manifold
also can be used for systems in which a chemical re- action generates the species
responsible for the
analytical signal. In this case
the car- rier stream both
transports the sample to the detector and reacts with the sample.
Because the sample
must mix with
the carrier stream,
flow rates are
lower than when no chemical reaction
is involved. One example is the determination of chlo- ride in water, which
is based on the following sequence of reactions.
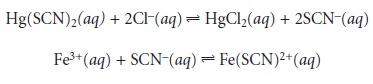
The carrier stream consists of an acidic solution of Hg(SCN)2 and Fe3+. When a
sample containing chloride is injected into
the carrier stream,
the chloride displaces the thiocyanate from Hg(SCN)2. The displaced thiocyanate then reacts with Fe3+ to form the reddish colored
Fe(SCN)2+ complex, the absorbance of which is moni-
tored at a wavelength of 480 nm. Sampling rates
of approximately 120 samples/h
have been achieved with this
system.
Most flow injection analyses requiring a chemical reaction
use a manifold containing more than one channel.
This provides more control over the mixing of reagents and the
interaction of the
reagents with the
sample. Two configura- tions are possible for dual-channel systems.
The dual-channel manifold
shown in Figure 13.22a
is used when
a mixture of the reagents is unstable. For
example, in acidic solutions
phosphate reacts with molybdate to form the heteropoly acid H3P(Mo12O40). In the presence of ascorbic acid the molybdenum in the het- eropoly acid is reduced
from Mo(VI) to Mo(V), forming
a blue-colored complex that is monitored spectrophotometrically at 660 nm.
Solutions of molybdate and ascorbic acid are maintained in separate reservoirs because they are not sta- ble
when mixed together. The two reagent
channels are merged
and mixed just before the point where the sample is injected.
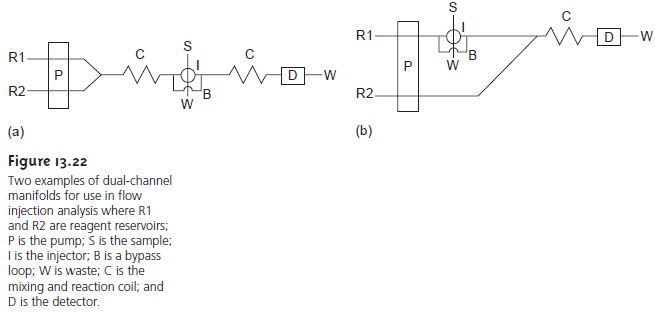
A dual-channel manifold can also be used to mix a reagent in a secondary chan- nel with a sample that has been injected into the primary
channel (Figure 13.22b). This style of manifold
has been used for the quantitative analysis
of many analytes, including the
determination of chemical oxygen demand (COD)
in wastewater.24 The COD
is a measure of the
amount of oxygen
required to completely oxidize the
organic matter in the water
sample and is an indicator of the level
of organic pollu- tion. In the conventional method of analysis, COD is determined by refluxing the sample for 2 h in the presence of acid and a strong
oxidizing agent, such as K2Cr2O7
or KMnO4. When refluxing is complete, the amount of oxidant consumed
in the re- action is determined by a redox
titration. In the flow injection version of this analy-
sis, the sample is injected into a carrier
stream of aqueous
H2SO4, which merges
with a solution of the oxidant
from a secondary channel. The oxidation reaction is kineti- cally slow. As a result, the
mixing and reaction coils are very
long, typically 40 m, and are
submerged in a thermostated bath.
The sampling rate is lower
than that for most
flow injection analyses, but at 10–30 samples/h
it is substantially greater than that for the conventional method.
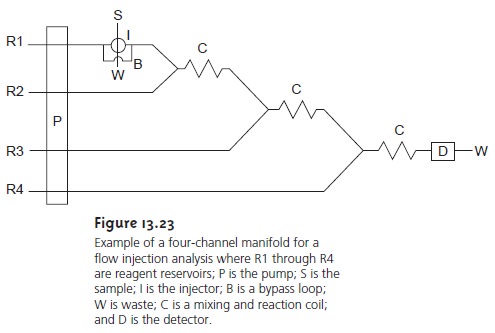
More complex manifolds
involving three or more channels
are common,
but the possible combination of designs is too numerous
to discuss in this text.
One ex- ample of a four-channel manifold is shown
in Figure 13.23.
Separation Modules
Incorporating a separation
module in the flow injection
manifold allows separations, such as dialysis, gaseous diffusion, and liquid–liquid
extraction, to be included in a flow injection analysis. Such separations are never
complete, but are reproducible if the operating conditions are carefully controlled.
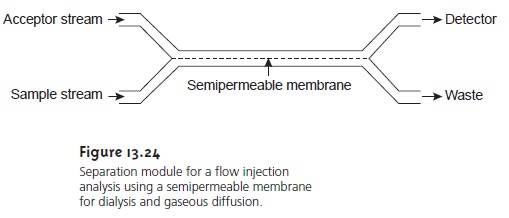
Dialysis and gaseous diffusion are accomplished by placing a semipermeable membrane between
the carrier stream
containing the
sample and an acceptor
stream (Figure 13.24).
As the sample stream passes through the separation mod- ule,
a portion of those species
capable of crossing
the semipermeable membrane do so, entering the
acceptor stream. This
type of separation module is common
in the analysis of clinical
samples, such as serum and urine, for which dialysis separates the analyte from its
complex matrix. Semipermeable gaseous diffusion membranes have been used for the determination of ammonia and carbon diox- ide in blood. For example, ammonia
is determined by injecting the sample into a
carrier stream of aqueous NaOH.
Ammonia diffuses across
the semipermeable membrane into an acceptor
stream containing an acid–base indicator. The re- sulting acid–base reaction between ammonia and the indicator is monitored
spectrophotometrically.
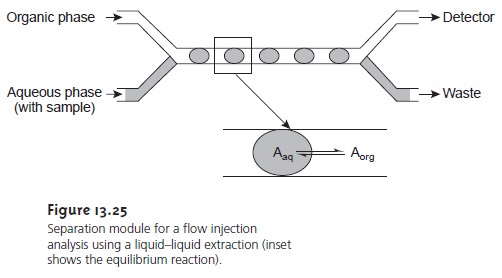
Liquid–liquid extractions are
accomplished by merging
together two immis-
cible fluids, each carried in a separate
channel. The result
is a segmented flow through
the separation module, consisting of alternating portions of the two phases. At the outlet
of the separation module, the two fluids
are separated by taking advantage of the
difference in their
densities. Figure 13.25
shows a typical configuration for a separation module in which
the sample is injected into an
aqueous phase and extracted into a less dense organic
phase that passes
through the detector.
Related Topics