Chapter: Modern Pharmacology with Clinical Applications: Drug Metabolism and Disposition in Pediatric and Gerontological Stages of Life
Drug Disposition in Pediatric Patients
DRUG DISPOSITION
IN PEDIATRIC PATIENTS
In spite of recent advances
in this area, knowledge of the disposition and actions of drugs in children is
lim-ited. This lack of information has made drug therapy for them difficult and
dangerous. There are two major ob-stacles to clinical drug studies in children.
One is an eth-ical issue, the inability to obtain true informed consent. The
second obstacle is inherent to children; they grow and change rapidly. Drug
studies must be performed on children at each stage of their development to
deter-mine appropriate usage for all patients.
To study drug disposition in
children it is most in-formative to divide them into five age groups: preterm
infants, term infants from birth through the first month of life, children 1
month to 2 years of age, children 2 to12 years of age, and children 12 to 18
years of age. Tanner staging of sexual maturation may more appro-priately break
down this latter group. Children that are Tanner stages I, II, and III are
appropriately considered children; those who are Tanner stages IV and V are
con-sidered adults.
Preterm infants, especially
those near the limits of viability (24 weeks’ gestation), have glomerular
filtration rates approximately one-tenth that of a term newborn. Because of
limitations on tubular reabsorption, they have increased urinary loss of
filtered substances. Glucuronidation pathways appear after 20 weeks of
ges-tation and so are limited in extremely premature infants.
Recent advances have made it
possible for drug therapy to begin prior to birth. Many mothers and therefore
their infants are receiving corticosteroids to induce maturation of the lungs.
Some fetal cardiac ar-rhythmias, such as supraventricular tachycardia, are
suc-cessfully managed by treating the mother during preg-nancy. Since most
drugs cross the placenta, the infant has the potential to be affected by drugs
that the mother takes. Metabolism and excretion are not the re-sponsibility of
the fetus, as the placenta and the mater-nal liver and kidneys contribute
significantly to drug elimination.
At birth, term infants can
metabolize and eliminate drugs. For most patients these systems did not function
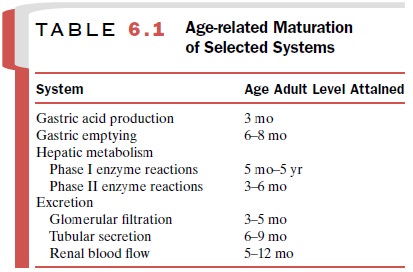
during fetal life and therefore even at birth are not very efficient. Table 6.1
outlines the time required for matu-ration of some of the systems used in drug
absorption and elimination. Table 6.2 lists other factors that alter drug
disposition in newborns. The ability to absorb and eliminate drugs increases
slowly over the first month of life.

Maternally administered drugs
also may affect in-fants who are breast-fed. Most drugs are present in breast
milk in small quantities. However, several drugs can reach concentrations
sufficient to adversely affect the newborn. Drugs that are contraindicated
during breast-feeding include cocaine, ergotamine, and cimeti-dine.
Unfortunately, for many drugs the information re-garding risks to the infant
from drug in breast milk is not available.
The period from 1 month to 2
years of age is a time of rapid growth and maturation. By the end of this
pe-riod, most systems function at adult levels. Paradoxically, between 2 and 12
years of age drug clearance greatly in-creases and often exceeds adult levels.
Half-lives are shorter and dosing requirements are frequently greater than for
adults (Table 6.3).

From 12 to 18 years of age
sex differences start to appear. These differences are often associated with a
decreased drug absorption and elimination in the fe-male as opposed to the
male. Females have less gastric acidity and an increased gastric emptying time.
Estro-gens decrease hepatic cytochrome P450 content and therefore may decrease
metabolism of some drugs via phase I pathways. Cyclic changes in glomerular
filtra-tion are noted during the menstrual cycle.
Absorption
Oral absorption of drugs is influenced by gastric acidity and emptying time. Gastric acid is rarely found in the stomach of infants at less
than 32 weeks’ gestation. Acid initially is secreted within the first few hours
after birth, reaching peak levels within the first 10 days of life. It
de-creases during the next 20 days of extrauterine life. Gastric acid secretion
approaches the lower limits of adult values by 3 months of age. The initiation
of acid secretion is often delayed in infants with delayed initia-tion of oral
feedings, such as extreme preemies and those with anomalies of the
gastrointestinal tract.
Gastric emptying time in
infants is related to their age and to the type of formula they receive.
Formulas containing long-chain fatty acids will delay gastric emp-tying. Both
gastric emptying time and small-intestine peristalsis tend to be slow until the
later part of the first year of life. In children aged 2 to 12 years gastric
emp-tying time dramatically increases, as does splanchnic blood flow. These
physiological changes result in faster drug absorption and increased peak blood
concentra-tions of drug. The decreased small intestine transit time during this
period may result in decreased absorption of some drugs. Because of low blood
flow through muscles in the neonatal period, drugs administered
intramuscu-larly are absorbed erratically.
Percutaneous drug absorption
can present special problems in newborns, especially in preterm infants. While
the skin of a newborn term infant may have the same protective capacity as the
skin of an adult, a preterm infant will not have this protective barrier until
after 2 to 3 weeks of life. Excessive percutaneous ab-sorption has caused
significant toxicity to preterm ba-bies. Absorption of hexachlorophene soap
used to bathe newborns has resulted in brain damage and death. Aniline dyes on
hospital linen have caused cyanosis secondary to methemoglobinemia, and EMLA
(lidocaine/prilocaine) cream may cause methemoglo-binemia when administered to
infants less than 3 months of age.
Distribution
The total body water of
prematures, newborns, and in-fants is significantly greater than it is for
older children and adults. This increased total body water increases the volume
of drug distribution for water-soluble com-pounds. As a consequence, there is a
need to administer loading doses of some drugs. Differences in total body water
are basically insignificant after the first year of life. Newborns have
decreased body fat and therefore less storage ability for fat-soluble drugs.
Newborns, especially
prematures, have decreased plasma albumin and total plasma protein
concentrations. In addition, albumin from these patients shows a de-creased
drug-binding affinity. This may result in increased plasma levels of free drug
and the potential for toxicity. In the past, concerns were raised that certain
drugs, such as sulfonamides, could displace endogenous substances, like
bilirubin, from albumin-binding sites. Theoretically, such an interaction would
increase the risk for kernicterus. Although this belief has been challenged
recently, reluc-tance to treat newborns with sulfonamides persists.
Metabolism
As with adults, the primary
organ responsible for drug metabolism in children is the liver. Although the
cy-tochrome P450 system is fully developed at birth, it functions more slowly
than in adults. Phase I oxidation reactions and demethylation enzyme systems
are signif-icantly reduced at birth. However, the reductive enzyme systems
approach adult levels and the methylation pathways are enhanced at birth. This
often contributes to the production of different metabolites in newborns from
those in adults. For example, newborns metabolize approximately 30% of
theophylline to caffeine rather than to uric acid derivatives, as occurs in
adults. While most phase I enzymes have reached adult levels by 6 months of
age, alcohol dehydrogenase activity appears around 2 months of age and
approaches adult levels only by age 5 years.
Phase II synthetic enzyme
reactions are responsible for the elimination of endogenous compounds, such as
bilirubin, and many exogenous substances. The immatu-rity of the
glucuronidation pathway was responsible for the development of gray baby
syndrome (Preterm and newborn infants dying of this syndrome developed anemia
and cardiovascular collapse because of high blood concentrations of
unconjugated chlorampheni-col. The plasma half-life was found to be 26 hours in
these patients compared with 4 hours in older children.
Infants and children have a
greater capacity to carry out sulfate conjugation than do adults. For example,
acetaminophen is excreted predominantly as a sulfate conjugate in children as
opposed to a glucuronide con-jugate in adults. This enhanced sulfation of
acetamino-phen, along with decreased metabolism via cytochrome P450 pathways
and increased glutathione turnover, are thought to explain the decreased
hepatotoxicity caused by this analgesic in children under 6 years of age. Phase
II enzyme systems reach adult levels between 3 and 6 months of age.
Excretion
Renal blood flow, glomerular
filtration rate, and tubular function are reduced in both preterm and term
neonates. Therefore, newborns, especially those less than 34 weeks’ gestation,
require less frequent dosing inter-vals for many drugs. Aminoglycosides are
administered every 8 hours in older children, every 12 hours in new-borns, and
every 24 hours in extremely premature in-fants. The glomerular filtration rate
of the term newborn is approximately 50% less than the adult level but reaches
adult values by 1 year of age. Renal blood flow approaches adult values between
ages 5 and 12 months. Tubular secretory functions mature at a slower rate than
does glomerular filtration. Renal excretion of organic anions, such as
penicillin, furosemide, and indomethacin, is very low in the newborn. Tubular
secretion and reab-sorption reach adult levels by 7 months of age. Renal
elimination of drugs appears to play a greater role than does metabolism in
newborns. Over the first year of life the infant develops a more adult-type
excretory pattern.
Drug Action
Most drugs are administered
to infants and children for the same therapeutic indications as for adults.
However, a few drugs have found unique uses in children. Among these are
theophylline and caffeine, which are used to treat apnea of prematurity;
indomethacin, which closes a patent ductus arteriosus; and prostaglandin E1,
which maintains the patency of the ductus arteriosus. Para-doxically, drugs
such as phenobarbital, which have a sedating action on adults, may produce
hyperactivity in children, and some adult stimulant drugs, such as
methyl-phenidate, are used to treat children with hyperactivity.
Adverse Reactions
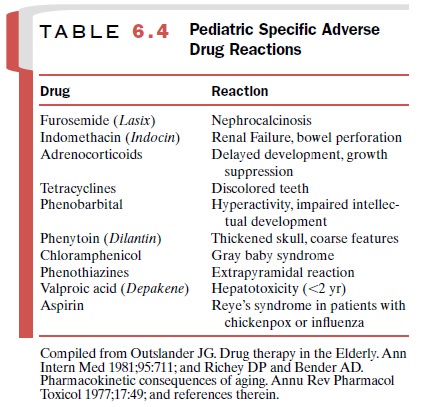
Children may display adverse
reactions different from those noted in adult patients. Table 6.4 lists a
number of drugs that demonstrate unique actions in children.
Special Considerations
Several problems unique to
pediatric drug therapy de-serve special mention. For example, most medications
are commercially available only in adult dose forms. Preparing pediatric doses
from adult tablets or capsules can be very difficult and may require special
skill on the part of the pharmacist. For some drugs it is simpler to administer
the intravenous (IV) preparation orally than to develop a preparation from the
oral medication.
IV drug administration is most effective in children when given via a pump infusion system close to the site of IV insertion. Because of the small size of many pedi-atric doses and the fact that some drugs adhere to IV tubing, a significant percentage of the drug can be lost if it is given using techniques usually reserved for adults.
For many prematures and newborns, the volume of
ad-ministration is also critical and therefore much more easily managed by IV
infusion pumps.
Most adult drugs must be
diluted to achieve appro-priate pediatric dosages. Some drugs must be diluted
several times. This introduces the potential for signifi-cant error in
dilution. Some drugs such as NPH (Neutral Protamine Hagedorn) insulin may lose
their effective-ness if diluted.
Children with chronic
illnesses require special con-sideration. For example, patients with cystic
fibrosis have increased hepatic metabolism and therefore in-creased drug
clearance. This may necessitate the admin-istration of increased drug dosages.
Calculation of pediatric
dosages is usually done on the basis of weight (e.g., milligrams per kilogram)
for in-fants and toddlers and on the basis of weight or body surface area
(milligrams per square meter) for older children. Repeated increases in drug
dosage are re-quired to accommodate for growth in children receiving chronic
drug therapy.
In summary, children,
especially those in the first year of life, present significant pharmacological
chal-lenges. Drug administration must be tailored to meet the unique needs of
children at their varied stages of de-velopment. Special attention must be
given to unex-pected drug actions and adverse reactions in these pa-tients, who
are maturing at variable rates. When planning drug therapy for children, it is
important to remember:
·
Children are not small adults.
·
Infants are not small children
·
Newborns are not small infants.
·
Preemies are not small newborns.
Related Topics