Chapter: Clinical Cases in Anesthesia : Local Anesthetics
Describe the toxic effects of local anesthetics
Describe the toxic effects of local anesthetics.
Systemic toxicity of local anesthetics is the
result of the effect of excess plasma concentrations of these drugs on the
central nervous system and cardiovascular system. Most often, excess plasma
concentration of local anesthetic solution is due to accidental intravascular
injection. Less frequently, it may result from absorption of local anesthetic
solution from tissue injection sites. As blood levels increase, the central
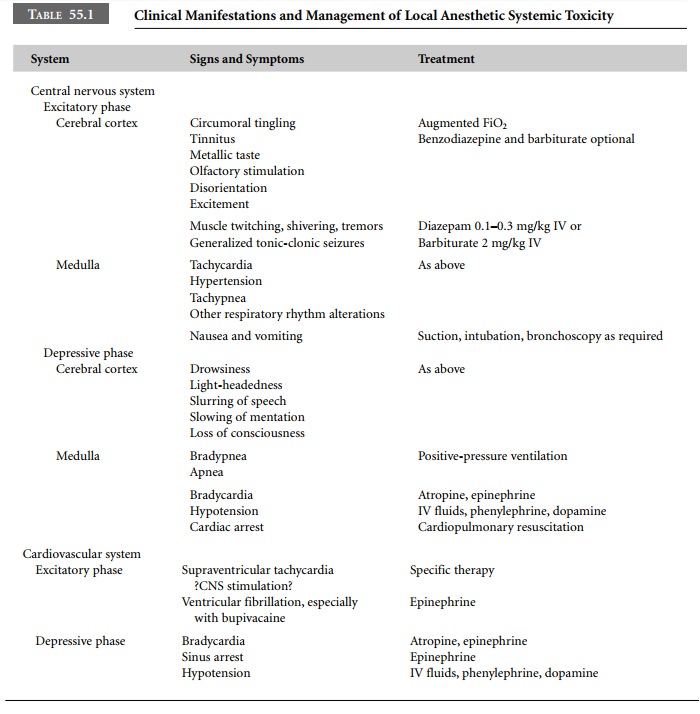
nervous system is affected first. Central nervous system manifestations of
local anesthetic toxicity are both excitatory and inhibitory in nature.
Increasing concentra-tions of local anesthetics in the blood initially depress
inhibitory neurons in the cerebral cortex allowing facilitatory neurons to
discharge without the normal negative input. Consequently, excitatory pathways
function in an unopposed manner, lead-ing to signs and symptoms of central
nervous system excita-tion (Table 55.1). Further increases in local anesthetic
blood levels depress excitatory neuronal activity, producing signs and symptoms
of central nervous system depression.
Acid–base status may also affect seizure
threshold. Respiratory acidosis tends to decrease the local anesthetic blood
concentration required to produce seizures. Elevated arterial carbon dioxide
tension (PaCO2) levels will increase cerebral blood flow, thereby
delivering additional local anesthetic to neurons. Diffusion of carbon dioxide
into neurons predisposes patients to intracellular acidosis and cationic
trapping. Positively charged forms of the local anesthetic tend to concentrate
within the neuron, thereby exerting a greater effect than would otherwise have
been expected at a particular blood concentration.
The cardiovascular system is not as vulnerable
to local anesthetic toxicity as the central nervous system. Local anesthetics
act directly on the heart by blocking cardiac Na+ channels, to
produce both electrophysiologic and
They decrease the rate of
depolarization in Purkinje fibers and ventricular muscle. Consequently, heart
rate decreases as automaticity and conduction slow, manifesting on the
electrocardiogram as P-R interval prolon-gation and QRS complex widening.
Profound hypotension from arteriolar vascular smooth muscle relaxation and
direct myocardial depression can occur. Ventricular dysrhythmias, including
ventricular fibrillation, may also manifest. Local anesthetics differ in their
ability to produce cardiac toxicity. The most potent local anesthetics,
bupivacaine, etidocaine, and tetracaine, affect rhythm and contractility at
lower blood concentrations than less potent agents. Bupivacaine has a greater
tendency to produce cardiac toxicity (selective cardiac toxicity) compared with
other agents because of the slow dissociation of highly lipid-soluble
bupivacaine from cardiac Na+ channels. This results in an
exaggerated and persistent depressant effect of bupivacaine on cardiac
function. The dextroisomer of bupivacaine is believed to be responsible for the
anesthetic’s systemic toxic effects. Thus, ropivacaine and levobupivacaine,
both pure levoisomers, are less cardiotoxic than bupivacaine. Bupivacaine seems
to be more cardiotoxic in pregnant patients. Resuscitation from
bupivacaine-induced cardiac arrest requires massive doses of epinephrine and
atropine.
Direct effects of local anesthetics on the
peripheral vasculature are variable. Central nervous system effects generally
predominate over direct vasodilating or vasocon-stricting influences.
Cocaine is unique among all local anesthetics
because it impairs reuptake of norepinephrine. The norepinephrine that is not
returned to storage granules produces vaso-constriction. Use of 10% cocaine has
been associated with coronary artery constriction and dysrhythmias. Four
percent cocaine functions well as a local vasoconstrictor without these
side-effects.
Prilocaine and benzocaine have been reported to
pro-duce methemoglobinemia. Prilocaine at doses higher than 600 mg may result
in the accumulation of ortho-toluidine and nitro-toluidine, both of which are
capable of causing methemoglobin formation. Patients with methemoglo-binemia
may appear cyanotic and their blood may be chocolate-colored because of the
impaired oxygen transport. Treatment of significant methemoglobinemia requires
intra-venous administration of methylene blue.
In addition to systemic toxicity, the use of
local anes-thetics for spinal and epidural anesthesia may result in transient
radicular irritation or overt neurotoxicity as manifested by the cauda equina
syndrome. Transient radic-ular irritation, the majority of cases occurring in
the lumbar and sacral areas, produces moderate to severe pain in the lower
back, buttocks, and posterior thighs that occurs within 24 hours after complete
recovery from spinal anesthesia. Full neurologic recovery usually occurs within
a week. Lidocaine, with concentrations ranging from 0.5% to 5%, seems to be
associated with a higher incidence of transient radicular irritation compared
with bupivacaine or tetracaine. Furthermore, the incidence seems to be
increased when the lithotomy position is used during surgery. Though the
etiology of the radicular irritation is still unknown, use of lidocaine doses
greater than 80– 100 mg for spinal anesthesia has been questioned.
Cauda equina syndrome has been reported
following cases that are believed to have concentrated subarachnoid lidocaine
and/or bupivacaine at the cauda equina. This syndrome was reported as a
complication of continuous spinal anesthesia with lidocaine 5% and the usage of
small microcatheters. Pooling of very high local anesthetic concentration on
dependent nerves is thought to be contributory. For this reason, microcatheters
were removed from the market. However, rare occurrences of this syndrome
continue to be reported despite the absence of micro-catheters. They may be due
to the accidental subarachnoid injection of the intended epidural dose, repeat
administra-tion of local anesthetic after a failed spinal anesthetic, or the
use of large doses of local anesthetic during continuous spinal anesthesia
without the use of microcatheters. The syn-drome presents with varying degrees
of sensory anesthesia, bowel and bladder sphincter dysfunction, and paraplegia.
Related Topics