Classsification and Uses | Matter Around Us | Chapter 9 | 8th Science - Compounds | 8th Science : Chapter 9 : Matter Around Us
Chapter: 8th Science : Chapter 9 : Matter Around Us
Compounds
Compounds
A compound is a pure substance which
is formed due to the chemical combination of two or more elements in a fixed
ratio by mass. The properties of a compound are different from those of its
constituents. Water, carbon dioxide, sodium chloride etc. are few examples of
compounds. A molecule of water is composed of one oxygen atom and two hydrogen
atoms in the ratio 1:2 by volume or 8:1 by mass.
Classification of
Compounds
Based on the origin of chemical
constituents, compounds are classified as inorganic compounds and organic
compounds.
a. Inorganic compounds
Compounds obtained from non living
sources such as rock, minerals etc. , are called inorganic compounds. Example:
Chalk, baking powder etc. ,
b. Organic compounds
Compounds obtained from living
sources such as plants, animals etc. , are called organic compounds. Example:
Protein, carbohydrates, etc. ,
Both inorganic and organic compounds
exist in all three states ie., solids, liquids and gases. Let us learn about
some important compounds in solid, liquid and gaseous states.
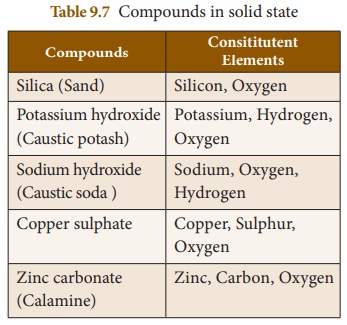
Some compounds that exist in solid
state are given in Table 9.7.
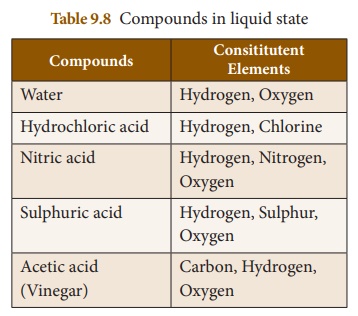
Compounds which exist in liquid
state are given in Table 9.8.
Some compounds exist in gaseous
state also. They are given in Table 9.9.

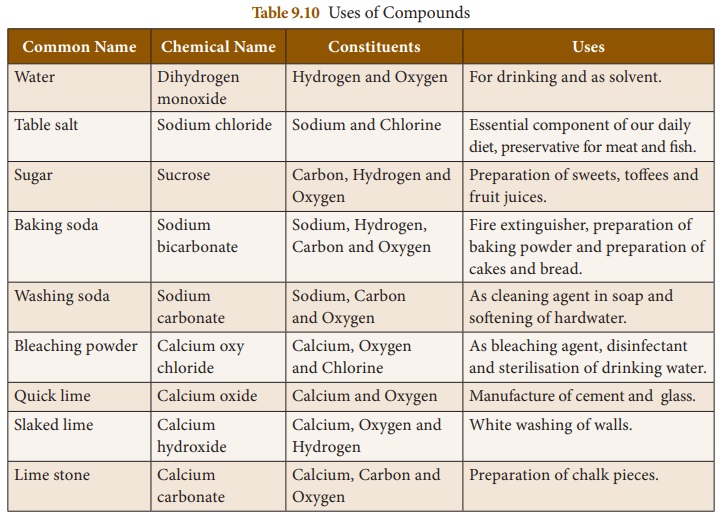
Uses of Compounds
We use a number of compounds in our
daily life. Some of them are listed in table 9.10.
Related Topics