Chapter: 9th Science : Carbon and its Compounds
Compounds of Carbon - Classification
Compounds of Carbon - Classification
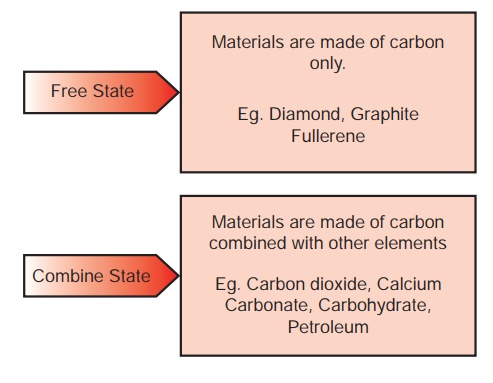
Carbon is found both in
free state as well as combined state in nature.
In the pre-historic
period, ancients used to manufacture charcoal by burning organic materials.
They used to obtain carbon compounds both from living things as well as
non-living matter. Thus in the early 19th century, Berzelius classified carbon
compounds based on their source as follows:
i. Organic Carbon Compounds: These are the
compounds of carbon obtained from living organisms such as plants and animals.
e.g. Ethanol, cellulose, Starch.
ii. Inorganic Carbon Compounds: These are the compounds
containing carbon but obtained from non-living matter. e.g. Calcium Carbonate,
Carbon Monoxide, Carbon dioxide.
1. Organic Compounds of Carbon
There are millions of
organic carbon compounds available in nature and also synthesized manually.
Organic carbon compounds contain carbon connected with other elements like
hydrogen, oxygen, nitrogen, sulphur etc. Thus depending on the nature of other
elements and the way in which they are connected with carbon, there are various
classes of organic carbon compounds such as hydrocarbons, alcohols, aldehydes
and ketones, carboxylic acids, amino acids, etc. You will study about organic
carbon compounds in your higher classes.
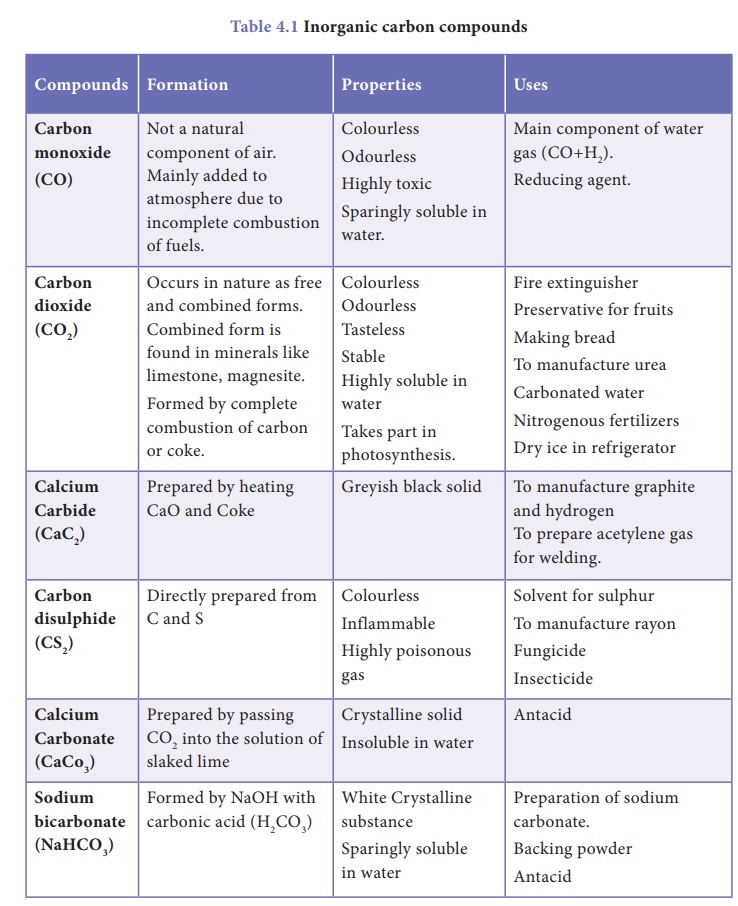
2. Inorganic Compounds of Carbon
As compared to organic
compounds, the number of inorganic carbon compounds are limited. Among them
oxides, carbides, sulphides, cyanides, carbonates and bicarbonates are the
major classes of inorganic carbon compounds. Formation, properties and uses of
some of the compounds are given in Table 4.1.
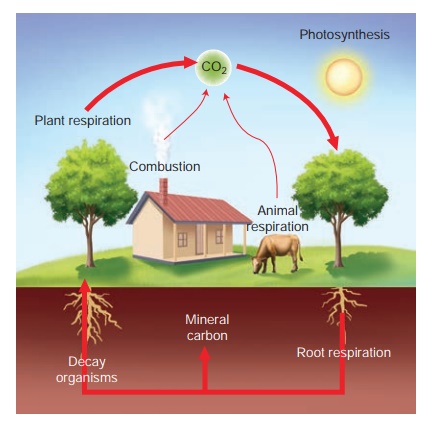
Carbon cycle
The carbon cycle is
the biogeochemical cycle by which carbon is exchanged among the biosphere,
geosphere, hydrosphere and atmosphere of the Earth. Carbon is the main
component of biological compounds as well as a major component of many minerals
such as limestone. Along with the nitrogen cycle and the water cycle, the
carbon cycle comprises a sequence of events that are key to make Earth capable
of sustaining life.
Related Topics