Chapter: Surgical Pathology Dissection : The Hematopoietic and Lymphatic System
Bone Marrow : Surgical Pathology Dissection
Bone Marrow
The
evaluation of the bone marrow typically in-cludes both a trephine core biopsy
and fluid aspi-ration. The details of the preparation of these specimens are
beyond the scope of this manual; however, you should be familiar with some
basics.
Trephine
core biopsies are usually 1.0 to 3.0 cm in length and 0.1 to 0.2 cm in
diameter. Because of their small size, they generally do not have to be
sectioned for further processing. After document-ing the size of the biopsy,
while it is still fresh prepare imprints from the biopsy by gently touching the
bony fragments to glass slides. These smears should be air dried for later
Wright’s staining or other special studies such as cyto-chemical and
immunohistochemical analyses. Next, submit the entire core biopsy for
process-ing. The choice of fixatives will depend on your individual
institution, but most laboratories like to fix bone marrow biopsies in either
Zenker’s fixative (for at least 4 hours), buffered formalin (for at least 18 to
24 hours), B-5 (for 4 hours), Bouin’s (for 4 to 12 hours), or a mixture of
buf-fered formalin and ethylenediaminetetraacetic acid (EDTA). Once fixed, the
specimen can be embedded in either paraffin or plastic. Although paraffin
embedding is certainly easier, plastic em-bedding has the advantage of
minimizing arti-facts produced by inadequate decalcification.
Usually,
approximately 1 cc of liquid is ob-tained from a bone marrow aspirate. Nine to
ten smears should be made immediately,
before the fluid clots. These smears can be prepared much the same way as
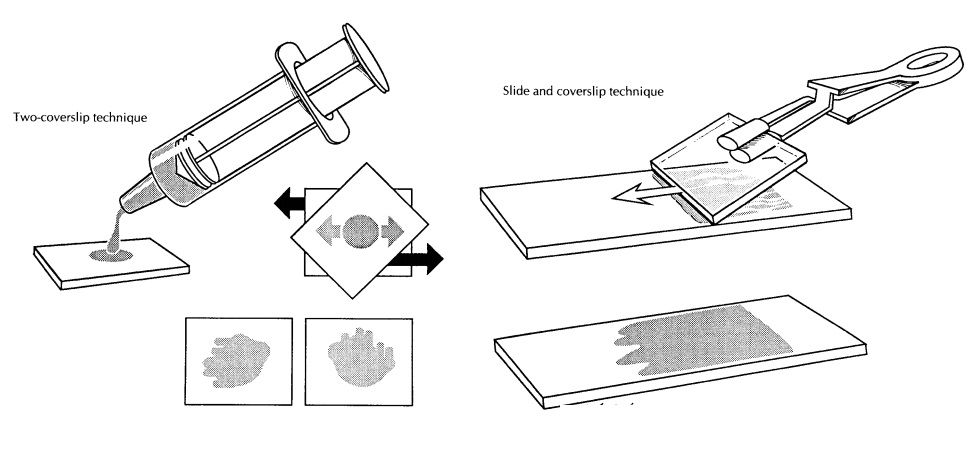
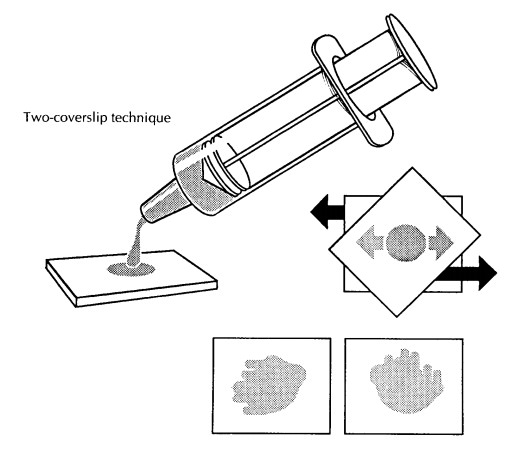
peripheral blood smears are made. To make smears on a coverslip, place a drop
on the edge of one coverslip, cover it with another coverslip placed at a
45-degree angle, and as the drop spreads pull the two coverslips apart
rapidly
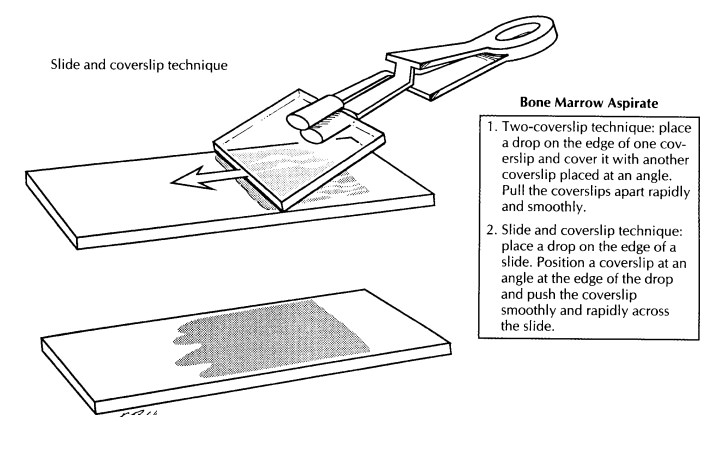
and smoothly. Alternatively, one drop of the aspirate can be placed at one end
of a glass slide and then gently smeared using a pusher coverslip, as
illustrated. These smears should be air dried. The remaining aspirate should be
placed in an anticoagulant such as EDTA and taken to the hematology laboratory
for further processing, which might include iron staining, the preparation of
additional smears, and possi-ble electron microscopy. Generally, additional
aspirates should be obtained for ancillary studies such as cytogenetics and
flow cytometry.
Finally,
excess aspirate fluid that has been al-lowed to clot can be submitted to the
laboratory for histologic processing after it has been fixed in formalin.
Important Issues to Address in Your Surgical Pathology Report on the Bone Marrow
• Describe
the cellularity, the relative numbers of myeloid and erythroid elements, and
their degree of maturation. The marrow biopsy can be used to assess
quantitative aspects of matu-ration, but subtle qualitative aspects are best
appreciated on the aspirate.
• Describe
the number of megakaryocytes, their arrangement, and any cytologic
abnormalities.
• Note the
degree and pattern of marrow fibrosis.
• Are
there any increases in lymphocytes or plasma cells (include the pattern of
infiltration and their cytology)?
• Is an
extrinsic tumor present?
• Is the
bone itself normal or abnormal?
Each of
these features should be commented on in the surgical pathology report,
although a very brief description can suffice for features that are normal.
Where possible, information from the biopsy, aspirate, and other ancillary
studiesshould be combined to make a definitive diag-nosis. In cases for which
only a biopsy is avail-able, sometimes only a descriptive diagnosis is given.
This practice varies from institution to institution.
Related Topics