Chapter: Aquaculture Principles and Practices: Sea-Basses and Sea-Breams
Asian sea-bass

Sea-basses
The main handicap to the introduction of intensive culture of the European sea-bass Dicentrarchus labrax and the Asian sea-bass Lates calcarifer has been the lack of dependablesource of fry and fingerlings. Though wild fry can be collected from natural habitats, the supply is highly inconsistent and inadequate. Because of this, many recent scientific studies on these species have been focused on developing methods of induced spawning and larval rearing.
Asian sea-bass
The Asian sea-bass Lates calcarifer, (fig. 23.5), which occurs in the tropical and sub-tropical areas of Asia, is a highly euryhaline species that lives in brackish-water estuaries and in fresh

For spawning they seem to require saline water, but larvae occur in fresh waters, including rice fields. The adult sea-bass is a voracious carnivore, but juveniles are omnivorous. One of the major problems in culturing them in ponds is their cannibalistic habits.
Induced spawning and larval rearing
In nature, L. calcarifer spawns all the year round, with the peak season from April to August. Though it does not spawn normally in confined areas, methods of induced spawning have been developed. Brood fish can be obtained from culture ponds or from open waters and reared in special earthen brood ponds, cement tanks or floating cages. It is reported that males predominate among smaller size groups (1.5–2.5kg body weight), but three to four year-old fish from culture ponds show a normal distribution of sexes (Kungvankij et al., 1986).Three-year-old females, weighing 3.5–5kg, and two-year-old males, weighing about 2.5–5kg, are preferred for artificial spawning. Spawning can be carried out in concrete tanks (of about 150 ton capacity) with a suitable supply of saline water (salinity of about 28–32ppt), with periodic water exchange and aeration.
The brood fish are introduced into the spawning tanks at the rate of about 10–12 pairs per tank, at least one month before the spawning. The mature female is recognized by the red-pink papilla extending out at the urinogenital aperture and the soft belly. The male is usually more slender, with slightly curved snout and, when mature, milt oozes out on slight pressure on the abdomen. Females with oocytes of about 0.5mm diameter are suitable for induced spawning.
The hormones usually used to induce spawning are HCG with pituitary gland of carp and Puberogen. Puberogen contains 63 per cent follicle stimulating hormone (FSH) and 34 per cent leutinizing hormone (LH). The spawners are usually given two intra-muscular injections at the base of the pectoral fin, the first one of 50IU HCG and 0.5–1 pituitary gland, and the second after 12 hours, of 100–200IU HCG and 1.5–2 pituitary glands. Within about 10– 12 hours after the second infection, spawning occurs. Repeated spawnings occur in batches over a period of three to five days. The fecundity ranges from 2–17 million eggs, depending on the size of the spawner. Fertilized eggs float on the surface of the tanks and can be siphoned out for hatching.
If the brood fish do not spawn in the tank after the second infection, they are stripped and the eggs artificially fertilized.
When Puberogen is used to induce spawning, the dosage is usually 50–200IU/kg body weight for the female and 20–25IU/kg fish for the male. If spawning does not occur within 36 hours, a second injection is given and the dosage doubled. This leads to spawning within 12–15 hours.
Induced spawning by environmental manipulation involves changing the salinity and temperature, simulating conditions in the natural spawning areas during the lunar phases. The salinity in the brood fish tank is gradually increased from 20–25ppt to 30–32ppt after the spawners are stocked, simulating the increased salinity to which the fish are exposed during migration from coastal areas to the sea. The pre-spawning behaviour of the spawners is carefully monitored. Segregation of the sexes may be done about a week before spawning. Spawning normally takes place during the full moon and new moon periods. At this time, the temperature of the water in the spawning tank containing the females and males is raised to 31–32°C by lowering the water level to about 30cm and exposing the water to the sun for two to three hours. Then the water temperature is suddenly lowered to 27–28°C by the addition of
filtered sea water. This induces the fish to spawn during the succeeding night. In case of failure the procedure is repeated. The fish usually spawn intermittently for about three to seven days.
The fertilized eggs are incubated in 50l capacity hatching jars or fibreglass tanks. Such containers can hold 50000–100000 eggs. A one-minute bath in 5ppm acriflavine followed by repeated rinsing in salt water is recommended before the eggs are introduced for hatching. The best hatching rates have been observed in salinities between 20 and 30ppt. With proper aeration and salinity, the eggs hatch out in about 17–18 hours at temperatures of 26–28°C. The hatchlings are about 1.5mm in length.
The hatchlings can be reared in large nursery tanks, supplied with water of about 20ppt salinity. The stocking density varies with the age and size of the larvae. The density is gradually decreased from 40000–50000 perm3 during the first week to 2000–5000 per m3 in the fourth week.
Experience indicates that sea-bass larvae require live food in their early stages. In hatcheries, the first food given to three-day-old larvae consists mainly of rotifers (Brachionusplicatilis), with a small percentage of Chlorella sp. and Tetraselmis sp., at the rate of 5–10 per ml. This may continue until the fourteenth day, with the addition of Artemia from the eighth day to the twentieth day. The suggested density of Artemia is 1 or 2 per ml. Usually the larval density in the tanks is reduced to about 20– 40 larvae/l, about a week after feeding starts. From the sixteenth day Daphnia or Moina can be added, at a density of 1 or 2 per ml, several times a day. After about three weeks, the fry are fed on minced fish. Generally the fry are graded during rearing to separate out the fast-growing ones from the others. Sorting and separation of fry according to size and thinning of stock help in reducing cannibalism among the fry. After about a month, the fry attain a size of about 12mm and are then used for grow-out in production ponds or cages.
Grow-out
Lates calcarifer has been cultivated for manyyears in brackish-water ponds, and in recent years in floating cages, but there is a lack of documented information on growout practices.
The main problem of grow-out are feeding and prevention of cannibalism among young fish. In order to reduce losses due to cannibalism, grow-out is performed in two phases. In the first phase the fry are grown to a weight of about 20g in special nursery-type ponds of up to 2000m2. Fry are stocked at the rate of 20–30/m2. Besides the natural food produced by fertilization, the fry are fed with supplementary feed consisting of adult Artemia and ground trash fish twice a day. Exchange of water at the rate of 30 per cent daily is maintained. The rearing period is about 30–45 days. By frequent sorting, fish of similar size are separated and stocked in separate grow-out facilities for growing to market size.
Grow-out to market size lasts for three to four months in countries like the Philippines, where 300–400g fish are acceptable, and 8– 12 months in other countries where 700–1200g fish are preferred. Floating and stationary cages of different sizes (usually 50m3) are used. The stocking density in the cages is about 40– 50 fish/m3, but after a growth of about three months the stock is thinned out to 10–20 fish/ m3. The usual feed is chopped trash fish, fed twice daily at the rate of 10 per cent of body weight initially, which is reduced to only once a day at 50 per cent of the body weight after about two months. When insufficient trash fish is available, rice bran or broken rice is added as a partial substitute.
Both monoculture and polyculture of sea-bass are practised. In intensive polyculture with the sea-bass as the main species, the subsidiary species are forage fish like tilapia. In such polyculture, the ponds are first stocked with the forage fish, which reproduces rapidly. When a sufficient stock of fry and juveniles of the forage has developed in the pond, sea-bass juveniles are stocked at the rate of 3000–5000 per ha. In monoculture systems the stocking rate is usually 10000–20000 per ha of uniform-sized juveniles which are fed daily with trash fish.
In traditional ponds, the sea-bass attain sizes around 500g in about 12 months. A gross production of about 2.76 tons/ha in eight months has been reported (Jhingran, 1977). It has been estimated that in monoculture in ponds with multiple stocking and harvesting, a production of about 3.3 tons/ha can be obtained.
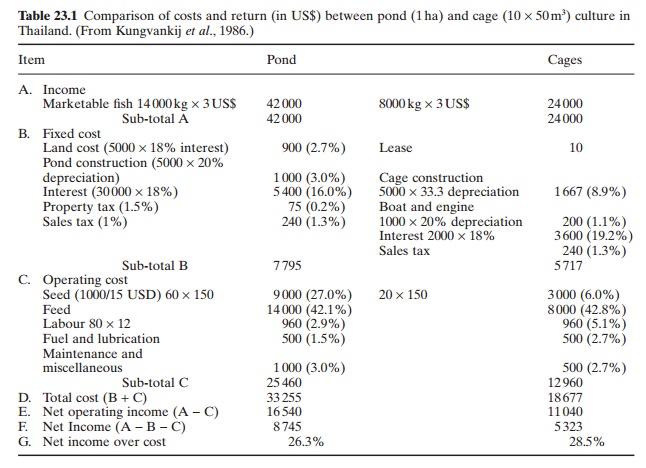
Intensive grow-out of sea-bass has proved to be economical, as can be seen from Table 23.1 which compares the costs and earnings of pond and cage culture in private farms in Thailand.
Asian sea-bass (barramundi) culture is being practised in Australia. The farming is done in cages, ponds and tanks. Cages of 1–300m3 in size are used, where 15–60kg/m3(25kg/m3 is optimal) of fish by mass is stocked. Tanks used are 10–30m3 and ponds 0.08–2ha in size. Until reaching 15cm total length, weekly size grading is done and full and frequent feeding adopted to avoid cannibalism during the weaning period. The stocked fish grow to 0.5, 0.8 and 3kg in 6, 12 and 18–24 months respectively; the size achieved varies with temperature. Juveniles stocked in freshwater reservoirs grow even to 10kg in three years. Juveniles have been stocked in freshwater reservoirs in Queens-land since 1983 to help declining recreational fisheries and also increase the value of the under-utilised environment. Juveniles for stocking produced in ponds are used widely now for stocking in impoundments in Queensland (Tucker et al., 2002).
Related Topics