Chapter: Medical Microbiology: An Introduction to Infectious Diseases: Arthropod-Borne and Other Zoonotic Viruses
Arbo Virus Disease
ARBOVIRUSES
ARBOVIRUS DISEASE
Some arboviruses cause severe inflammation of the brain (encephalitis) with damage or destruction of neural cells that may be fatal or lead to permanent neurologic damage in survivors. Others, such as dengue viruses, can produce illnesses that range from mild flu-like symptoms to overwhelming shock with widespread hemorrhage into tissues. Still another, yellow fever virus, primarily attacks liver cells, leading to extensive destruction and sometimes fatal liver failure.
EPIDEMIOLOGY
Arboviruses of major importance in human disease are listed in Table 40–1 with sum-maries of their geographic distribution, the arthropod vectors that transmit them, and the usual disease syndromes that can result from infection.
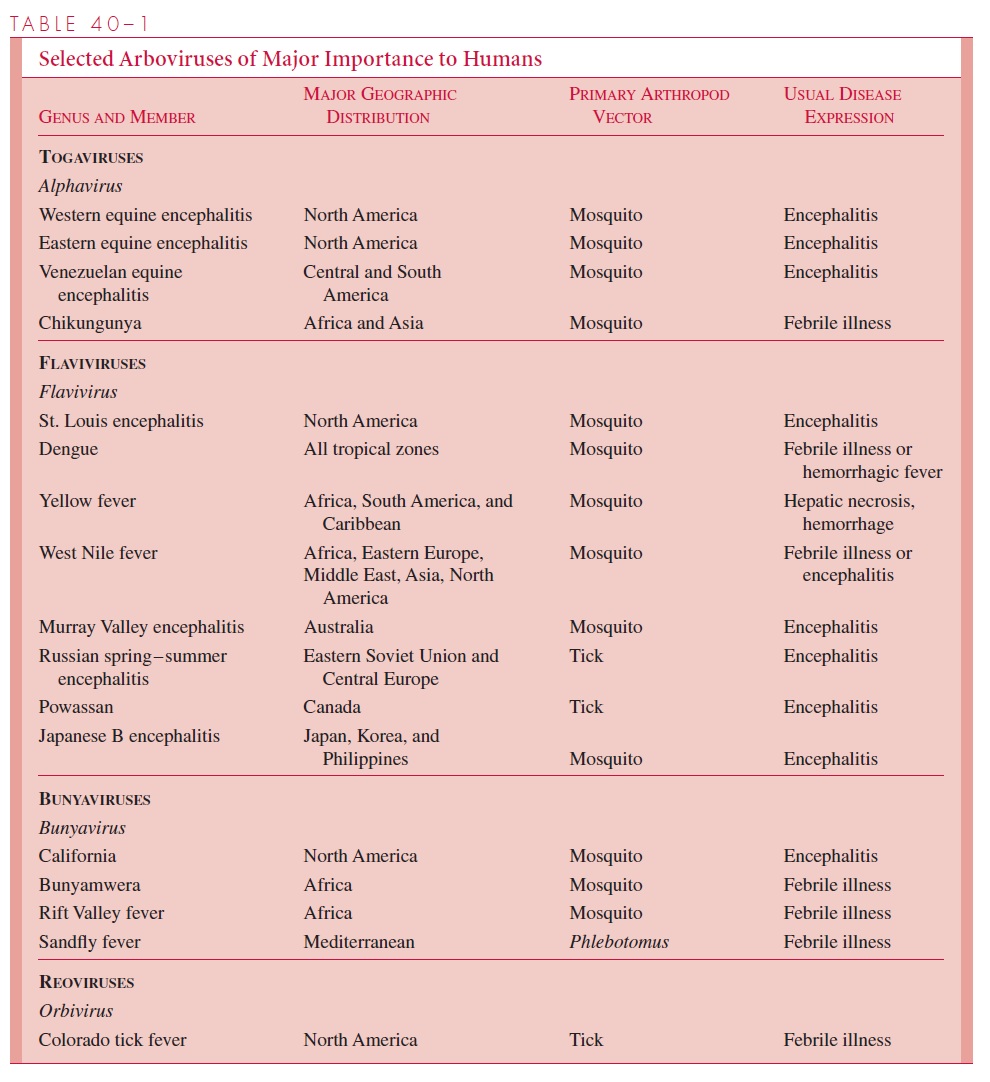
With the exception of urban dengue and urban yellow fever, in which the virus may simply be transmitted between humans and mosquitoes, other arboviral diseases involve nonhuman vertebrates. These are usually small mammals, birds, or, in the case of jungle yellow fever, monkeys. Infection is transmitted within the host species by arthropods (eg, mosquitoes or ticks) that become infected. In some cases, the infection can be maintained from generation to generation in the arthropod by transovarial transmission. Infection in the arthropod usually does not appear to harm the insect; however, a period of virus multi-plication (termed extrinsic incubation period) is required to enhance the capacity to transmit infection to vertebrates by bite. The consequences of infection transmitted from the arthropod to susceptible vertebrate hosts are variable; some develop illness of varying severity with viremia, whereas others may have long-term viremia without clinical disease. Vertebrate hosts are then a source of further spread of the virus by amplification, in which noninfected arthropods feeding on viremic hosts acquire the virus, thereby increasing the risk of transmission.
Transient viremia is a feature of many of these infections in hosts other than their reservoir; those affected, including humans and higher vertebrates (eg, horses and cattle), are often referred to as blind-end hosts. In contrast, if viremia is sustained for longer peri-ods (eg, weeks to months in a variety of togavirus, flavivirus, and bunyavirus infections of lower vertebrates), the vertebrate host becomes highly important as a reservoir for contin-uing transmission. Viremia may last a week or more in human dengue and yellow fever infections, and humans may then serve as a reservoir in urban disease.
Obviously, the usual arthropod vectors are rarely present during all seasons. The ques-tion then arises as to how the arboviruses survive between the time the vector disappears and the time it reappears in subsequent years. Several mechanisms can operate to sustain the virus between transmission periods (often referred to asoverwintering): (1) sustained viremia in lower vertebrates such as small mammals, birds, and snakes, from which newly mature arthropods can be infected when taking a blood meal; (2) hibernation of in-fected adult arthropods that survive from one season to the next; and (3) transovarial transmission, whereby the infected female arthropod can transmit virus to its progeny.
The three basic cycles of arbovirus transmission are urban, sylvatic, and arthropod-sustained.
Urban
As the term suggests, the urban cycle is favored by the presence of relatively large num-bers of humans living in close proximity to arthropod (usually mosquito) species capable of virus transmission. The cycle is:
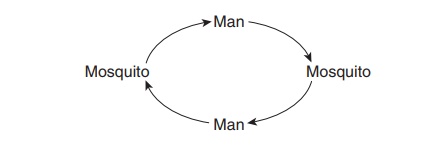
Examples of this cycle include urban dengue, urban yellow fever, and occasional urban outbreaks of St. Louis encephalitis.
Sylvatic
In the sylvatic cycle a single nonhuman vertebrate reservoir may be involved:
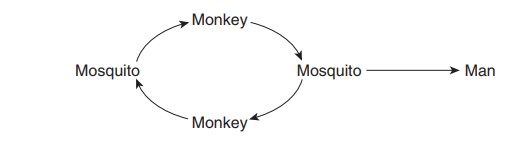
In this situation, the human, who becomes a tangential host through accidental intrusion into a zoonotic transmission cycle, is not important in maintaining the infection cycle. An example of this cycle is jungle yellow fever.
In other sylvatic cycles, multiple vertebrate reservoirs may be involved:
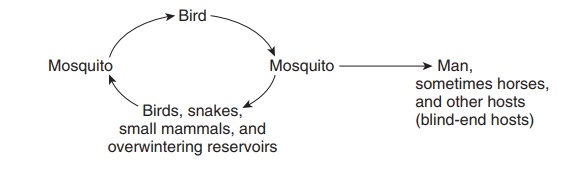
Examples include western equine encephalitis, eastern equine encephalitis, and California viruses. In some situations, such as St. Louis encephalitis and yellow fever, the urban and sylvatic cycles may operate concurrently.
Arthropod-Sustained
Arthropods, especially ticks, may sustain the reservoir by transovarial transmission of virus to their progeny, with amplification of the cycle by spread to and from small mammals:
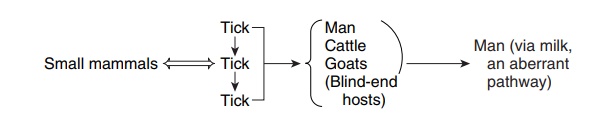
Tick-borne encephalitis in Russia is transmitted by this cycle. In temperate climates such as the United States, arboviruses are major causes of disease during the summer and early fall months, the season of greatest activity of arthropod vectors (usually mosquitoes or ticks). When climatic conditions and ecologic circumstances (eg, swamps and ponds) are optimal for arthropod breeding and egg hatching, arbovirus amplification may begin.
An example of amplification is provided by western equine encephalitis. When the mosquito vectors become abundant, the level of transmission among the basic reservoir hosts (birds and small mammals) increases, and the mosquitoes also turn to other suscep-tible species such as the domestic fowl. These hosts experience a rapidly developing asymptomatic viremia, which permits still more arthropods to become infected on biting. At this point, spread to blind-end hosts such as humans or horses and the development of clinical disease become likely. This occurrence depends on the accessibility of the host to the infected mosquito and on mosquito feeding preferences which, for unknown reasons, vary from one season to another.
PATHOGENESIS
There are three major manifestations of arbovirus diseases in humans associated with dif-ferent tropisms of various viruses for human organs, although overlap can occur. In some, the central nervous system (CNS) is primarily affected, leading to aseptic meningitis or meningoencephalitis. A second syndrome involves many major organ systems, with par-ticular damage to the liver, as in yellow fever. The third is manifested by hemorrhagic fever, in which damage is particularly severe to the small blood vessels, with skin petechiae and intestinal and other hemorrhages.
Infection of the human by a biting, infected arthropod is followed by viremia, which is apparently amplified by extensive virus replication in the reticuloendothelial system and vascular endothelium. After replication the virus becomes localized in various target organs, depending on its tropism, and illness results. The viruses produce cell necrosis with resultant inflammation which leads to fever in nearly all infections. If the major viral tropism is for the CNS, virus reaching this site by crossing the blood–brain barrier or along neural pathways can cause meningeal inflammation (aseptic meningitis) or neuronal dysfunction (encephalitis). The CNS pathology consists of meningeal and perivascular mononuclear cell infiltrates; degeneration of neurons with neuronophagia; and occasion-ally, destruction of the supporting structure of neurons.
In some infections, especially yellow fever, the liver is the primary target organ. Pathologic findings include hyaline necrosis of hepatocytes, which produces cytoplasmic eosinophilic masses called Councilman bodies. Degenerative changes in the renal tubules and myocardium may also be seen, as may microscopic hemorrhages throughout the brain. Hemorrhage is a major feature of yellow fever, largely because of the lack of liver-produced clotting factors as a result of liver necrosis.
Hemorrhagic fevers other than those related to primary hepatic destruction have a some-what different pathogenesis which has been studied most extensively in dengue infections. In uncomplicated dengue fever, which is associated with a rash and influenza-like symptoms, there are changes in the small dermal blood vessels. These alterations include endothelial cell swelling and perivascular edema with mononuclear cell infiltration. More severe infection, as in dengue hemorrhagic fever, often complicated by shock, is characterized by perivascular edema and widespread effusions into serous cavities such as the pleura and hemorrhages from the upper respiratory and intestinal tracts. The spleen and lymph nodes show hyperpla-sia of lymphoid and plasma cell elements, and there is focal necrosis in the liver. The patho-physiology seems related to increased vascular permeability and disseminated intravascular coagulation, which is further complicated by liver and bone marrow dysfunction (eg, de-creased platelet production, decreased production of liver-dependent clotting factors). The major vascular abnormalities may be provoked by circulating virus–antibody complexes (immune complexes) that mediate activation of complement and subsequent release of vasoactive amines. The precise reason for this phenomenon is not clear; it may be related to intrinsic virulence of the virus strains involved and to host susceptibility factors.
Two hypotheses are based on the existence of four distinct but antigenically related serotypes of dengue virus, any of which can generate group-specific cross-reacting anti-bodies that are not necessarily protective against other serotypes. One possibility is that preexisting group-specific antibody at a critical concentration serves as “enhancing” rather than neutralizing antibody. In the presence of enhancing antibody, virus–antibody complexes are more efficiently adsorbed to and engulfed by monocytes and macrophages. Subsequent replication leads to extensive spread throughout the host. Alternatively, or in concert with this, activation of previously sensitized T cells by viral antigen present on the surfaces of macrophages may result in release of cytokines, which mediate the devel-opment of shock and hemorrhage.
IMMUNITY
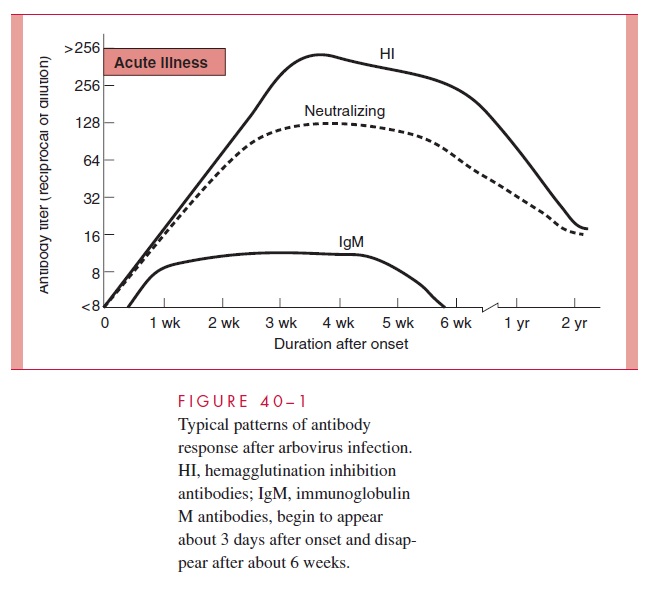
The usual humoral responses (hemagglutination inhibition, complement fixation, neutral-ization, precipitation) in relation to onset of illness are illustrated in Figure 40–1. The rise in antibody titer generally correlates with recovery from infection. Neutralizing antibodies, which are the most serotype specific, generally persist many years after infection. The presence of IgM-specific antibodies indicates that primary infection likely occurred within the previous 2 months. Cellular and humoral immunity to reinfection are serotype specific and appear to be permanent.
Related Topics