Chapter: Clinical Anesthesiology: Anesthetic Management: Geriatric Anesthesia
Age-Related Anatomic & Physiological Changes
Age-Related Anatomic & Physiological Changes
CARDIOVASCULAR SYSTEM
Cardiovascular diseases are more prevalent in
the geriatric than general population. Still, it is impor-tant to distinguish
between changes in physiology that normally accompany aging and the
pathophysi-ology of diseases common in the geriatric popula-tion (Table
43–2). For example, atherosclerosis is
pathological—it is not present in healthy elderly patients. On the other hand,
a reduction in arterial elasticity caused by fibrosis of the media is part of
the normal aging process. Changes in the cardiovas-cular system that accompany
aging include decreased vascular and myocardial compliance and autonomic
responsiveness. In addition to myocar-dial fibrosis, calcification of the
valves can occur. Elderly patients with systolic murmurs should besuspected of
having aortic stenosis. However, in the absence of co-existing disease,
restingsystolic cardiac function seems to be preserved, even in octogenarians.
Functional capacity of less than 4 metabolic equivalents (METS) is associated
with potential adverse outcomes (see Table 21–2). Increased vagal tone and
decreased sensitivity of adrenergic receptors lead to a decline in heart rate;
maximal heart rate declines by approximately one beat per minute per year of
age over 50. Fibrosis of the conduction system and loss of sinoatrial node
cells increase the incidence of dysrhythmias, partic-ularly atrial fibrillation
and flutter. Preoperative risk assessment and evaluation of the patient with
car-diac disease were previously reviewed in this text. Age per se does not
man-date any particular battery of tests or evaluative tools, although there is
a long tradition of routinely requesting tests such as 12-lead
electrocardiography

(ECG) in patients who are older than a defined age. Nonetheless, elderly
individuals are more likely to present for surgery with previously unde-tected
conditions that require an intervention, such as arrhythmias, congestive heart
failure, or myocardial ischemia. Cardiovascular evaluation should be guided by
American Heart Association guidelines.
Elderly
patients undergoing echocardio-graphic evaluation
for surgery have
an increased incidence of diastolic dysfunction com-pared with younger
patients. Diastolic dysfunction prevents the ventricle from relaxing and
conse-quently inhibits diastolic ventricular filling at rela-tively low
pressures. The ventricle becomes less compliant, and filling pressures are
increased. Dia-stolic dysfunction is NOT equivalent to diastolic heart failure.
In some patients, systolic ventricular function can be well preserved; however,
the patient can have signs of congestion secondary to severe diastolic
dysfunction. Diastolic heart failure mostoften coexists with systolic
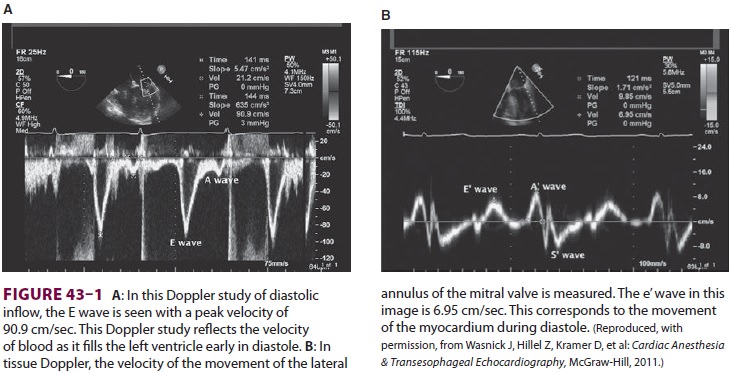
dysfunction. Echocardiography is used to assess diastolic dysfunction. A ratio of greater than 15 between the peak E velocity of
transmitral diastolic filling and the e’ tissue Doppler wave is associated with
elevated left ventricular end-diastolic pressure and diastolic dysfunction.
Conversely, a ratio of less than 8 is consistent with normal diastolic function
(see Figure 43–1).
Marked diastolic dysfunction may be seen with
systemic hypertension, coronary artery disease, car-diomyopathies, and valvular
heart disease, particu-larly aortic stenosis. Patients may be asymptomatic or
complain of exercise intolerance, dyspnea, cough, or fatigue. Diastolic
dysfunction results in relatively large increases in ventricular end-diastolic
pressure, with small changes of left ventricular volume; the atrial
contribution to ventricular filling becomes even more important than in younger
patients. Atrial enlargement predisposes patients to atrial fibrillation and
flutter. Patients are at increased risk of developing congestive heart failure.
The elderly patient with diastolic dysfunction may poorly toler-ate
perioperative fluid administration, resulting in elevated left ventricular
end-diastolic pressure and pulmonary congestion.Diminished
cardiac reserve in many elderly patients may be manifested as exaggerateddrops in blood pressure during induction of gen-eral anesthesia. A
prolonged circulation time delays
the onset of intravenous drugs, but speeds induc-tion with inhalational
agents. Like infants, elderly patients have less ability to respond to
hypovolemia, hypotension, or hypoxia with an increase in heart rate.
Ultimately, cardiovascular diseases, including heart failure, stroke,
arrhythmias, and hypertension contribute to an increased risk of morbidity,
mor-tality, increased cost of care, and frailty in elderly patients.
Research is ongoing into the relationship between telomere biology and cardiovascular dis-ease. Telomeres, which are located at the chromo-some terminus, protect the DNA from degradation during cell division. With each cell division, there is progressive telomere loss. Cells with short telo-meres undergo “replicative senescence” and apopto-sis. Telomerase maintains telomere length, but has low activity in human cells. Indeed, telomere length varies among humans based upon inheritance and environmental factors. Telomerase activity is defi-cient in various early aging syndromes. Telomere shortening may be either a cause or a consequence of cardiovascular disease. Whatever the exact mech-anism of cardiovascular aging, patient management should at all times be in accordance with American Heart Association/American College of Cardiology guidelines.
RESPIRATORY SYSTEM
Aging decreases the elasticity of lung
tissue, allowing overdistention of alveoli and collapseof small airways.
Residual volume and the func-tional residual capacity increase with aging.
Air-way collapse increases residual volume and closing capacity Even in normal
persons, closing capacity exceeds functional residual capacity at age 45 years
in the supine position and age 65 years in the sit-ting position. When this
happens, some airways close during part of normal tidal breathing, result-ing
in a mismatch of ventilation and perfusion. The additive effect of these
emphysema-like changes decreases arterial oxygen tension by an average rate of
0.35 mm Hg per year; however, there is a wide range of arterial oxygen tensions
in elderly preoper-ative patients. Both anatomic and physiological dead space
increase. Other pulmonary effects of aging are summarized in Table 43–2.
Decreased respiratory muscle function/mass, a
less compliant chest wall, and intrinsic changes in lung function can increase
the work of breathing and make it more difficult for elderly patients to muster
a respiratory reserve in settings of acute illness (eg, infection). Many
patients also present with obstruc-tive or restrictive lung diseases. In
patients who have no intrinsic pulmonary disease, gas exchange is unaffected by
aging.
Measures to prevent perioperative hypoxia in elderly patients include a
longer preoxygen-ation period prior to induction, increased inspired oxygen
concentrations during anesthesia, positive end-expiratory pressure, and
pulmonary toilet. Aspiration pneumonia is a common and poten-tially
life-threatening complication in elderly patients, possibly as a consequence of
a progressive decrease in protective laryngeal reflexes and immu-nocompetence
with age. Ventilatory impairment in the recovery room is more common in elderly
than younger patients. Factors associated with an increased risk of
postoperative pulmonary com-plications include age older than 64 years, chronic
obstructive pulmonary disease, sleep apnea, mal-nutrition, and abdominal or
thoracic surgical incisions.
METABOLIC & ENDOCRINE FUNCTION
Basal and maximal oxygen consumption declines with age. After reaching
peak weight at about age 60 years, most men and women begin losing weight; the
average elderly man and woman weigh less than their younger counterparts. Heat
production decreases, heat loss increases, and hypothalamic
temperature-regulating centers may reset at a lower level.
Diabetes affects approximately 15% of
patients older than age 70 years. Its impact on numerous organ systems can
complicate perioperative man-agement. Diabetic neuropathy and autonomic
dys-function are particular problems for the elderly.
Increasing insulin resistance leads to a
progres-sive decrease in the ability to avoid hyperglycemia with glucose loads.
Institutions typically have their own protocols on how to manage increased
blood glucose perioperatively, and these protocols reflect the changing
literature on “tight” control. Attempts to maintain blood glucose within a
strictly nor-mal range during surgery, anesthesia, and/or criti-cal illness may
lead to hypoglycemia and adverse outcomes. Anesthesia practitioners are advised
to determine what the “acceptable” perioperative blood glucose level is in
their institution and to be aware of changing performance benchmarks related to
this measure.
The neuroendocrine response to stress seems to be largely preserved, or,
at most, only slightlydecreased in healthy elderly patients. Aging is associated with a decreasing response toβ-adrenergic
agents.
RENAL FUNCTION
Renal blood flow and kidney mass (eg,
glomerular number and tubular length) decrease with age. Renal function, as
determined by glomerular filtration rate and creatinine clearance, is reduced
(Table 43–2). The serum creatinine level is unchanged because of a decrease in
muscle mass and creatinine produc-tion, whereas blood urea nitrogen gradually
increases with aging. Impairment of Na+ han-dling,
concentrating ability, and dilutingcapacity predispose elderly patients to both
dehy-dration and fluid overload. The response to antidi-uretic hormone and
aldosterone is reduced. The ability to reabsorb glucose is decreased. The
combi-nation of reduced renal blood flow and decreased nephron mass in elderly
patients increases the risk of acute renal failure in the postoperative period,
par-ticularly when they are exposed to nephrotoxic drugs and techniques.
As renal function declines, so does the
kidney’s ability to excrete drugs. The decreased capacity to handle water and
electrolyte loads makes proper fluid management more critical; elderly patients
are more predisposed to developing hypokalemia and hyper-kalemia. This is
further complicated by the common use of diuretics in the elderly population.
The search is ongoing for drugs that might protect the kidney peri-operatively,
as well as for specific genetic profiles of patients at greater risk of
perioperative kidney injury.
GASTROINTESTINAL FUNCTION
Liver mass and hepatic blood flow decline
with aging. Hepatic function declines in pro-portion to the decrease in liver
mass. Thus, the rate of biotransformation and albumin productiondecreases.
Plasma cholinesterase levels are reduced in elderly men.
NERVOUS SYSTEM
Brain mass decreases with age; neuronal loss
is prominent in the cerebral cortex, particularly the frontal lobes. Cerebral
blood flow also decreases about 10% to 20% in proportion to neuronal losses. It
remains tightly coupled to metabolic rate, and autoregulation is intact.
Neurons lose complexity of their dendritic tree and the number of synapses. The
synthesis of neurotransmitters, such as dopamine, and neurotransmitter
receptors are reduced. Sero-tonergic, adrenergic, and gamma-aminobutyric acid
(GABA) binding sites are also reduced. Astrocytes and microglial cells increase
in number.
Aging is associated with an increasing
threshold for nearly all sensory modalities, including touch, temperature
sensation, proprioception, hearing, andvision. Dosage requirements for local
and gen-eral (minimum alveolar concentration [MAC])
anesthetics are reduced. Administration of a
given volume of epidural local anesthetic tends to result in more extensive
spread in elderly patients. A longer duration of action should be expected from
a given dose of spinal local anesthetic.
Currently, much work is being done to
deter-mine whether surgery and anesthesia harm the brain in some manner.
Postoperative cognitive dys-function (POCD) is diagnosed by neurobehavioral
testing. Unlike delirium, which is a clinical diagno-sis, cognitive dysfunction
must be sought by using evaluative techniques. Up to 30% of elderly patients
can demonstrate abnormal neurobehavioral testing within the first week after an
operation; however, such testing may identify dysfunction already pres-ent in
these individuals prior to any surgery or anes-thesia exposure.
Ultimately, the question arises as to whether
general anesthetic agents result in neurotoxicity in the aged brain. Some
current investigations are attempting to determine whether anesthetic agents
produce POCD through a mechanism similar to that underlying Alzheimer’s
disease.
It is also possible that side effects of illness (eg, inflammation) and
the neuroendocrine stress response contribute to perioperative brain injury in
some manner, independent of anesthesia. Indeed, patients presenting for surgery
may pres-ent with cognitive dysfunction. In one study, 20% of elderly patients
presenting for elective total joint arthroplasty demonstrated preoperative
cognitive impairment; furthermore, POCD was independent of type of anesthesia
or surgery at 3 months post-operatively. Postoperative delirium is common in
elderly patients, especially those with reduced pre-operative neurocognitive
test scores and reduced functional status. Preoperative frailty is also
associated with postoperative delirium. Frailty is common in preoperative
elderly patients await-ing surgery and predicts postoperative delirium.
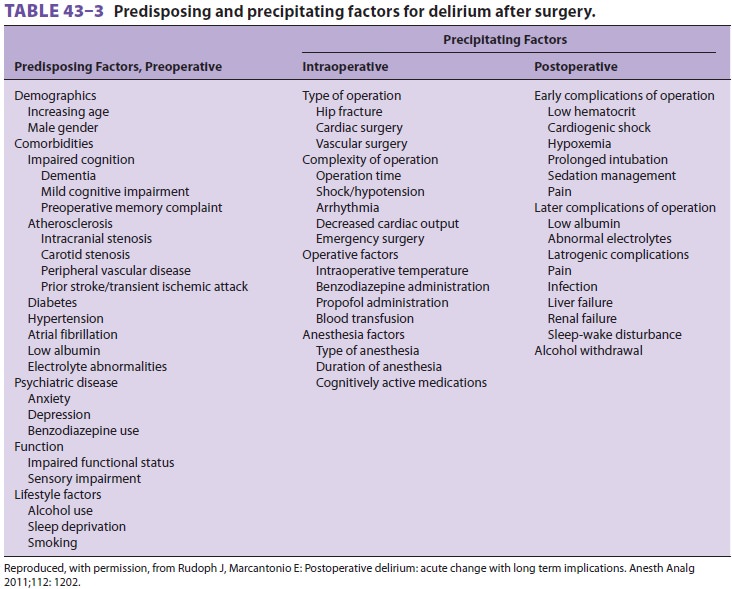
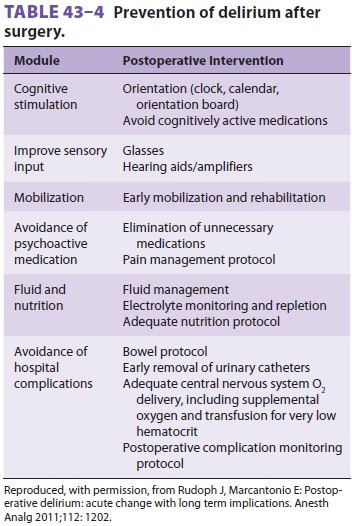
Delirium has a particularly frequent incidence fol-lowing hip surgery. Factors
associated with post-operative delirium in the elderly and ways to avoid it are
presented in Tables 43–3 and 43–4.
Elderly patients often take more time to recover completely from the
central nervous sys-tem effects of general anesthesia, especially if they were
confused or disoriented preoperatively. This is important in geriatric
outpatient surgery, where socioeconomic factors, such as the lack of a
care-taker at home, necessitate that patients may need to assume a higher level
of self care.
In the absence of disease, any perioperative decrease in cognitive
function is normally mod-est. Short-term memory seems to be most affected.
Continued physical and intellectual activity seems to have a positive effect on
preservation of cognitive functions.
The etiology of POCD is likely multifactorial and includes drug effects, pain, underlying dysfunc-tion, hypothermia, and metabolic disturbances. Elderly patients are particularly sensitive to centrally acting anticholinergic agents, such as scopolamine and atropine. Some patients suffer from prolonged or permanent POCD after surgery and anesthesia. Some studies suggest that POCD can be detected in 10% to 15% of patients older than age 60 years up to 3 months following major surgery. In some set-tings (eg, following cardiac and major orthopedic procedures), intraoperative arterial emboli may be contributory. Animal studies suggest that anesthe-sia without surgery can impair learning for weeks, particularly in older animals. Elderly inpatients seem to have a significantly higher risk of POCD than elderly outpatients. Anesthetic neurotoxic-ity is also a potential risk for the developing brain. Progress in research in this area is documented on the Smart TotsTM website (see http://www.smarttots.org/).
MUSCULOSKELETAL
Muscle mass is reduced in elderly patients.
Skin atrophies with age and is susceptible to trauma from removal of adhesive
tape, electrocautery pads, and electrocardiographic electrodes. Veins are often
frail and easily ruptured by intravenous infusions. Arthritic joints may
interfere with positioning or regional anesthesia. Degenerative cervical spine
dis-ease can limit neck extension, potentially making intubation difficult.
Related Topics