Chapter: Medical Physiology: Local and Humoral Control of Blood Flow by the Tissues
Acute Control of Local Blood Flow - Mechanisms of Blood Flow Control
Acute Control of Local Blood Flow
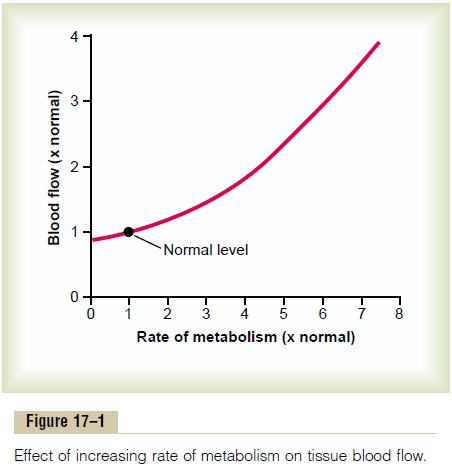
Effect of Tissue Metabolism on Local Blood Flow. Figure17–1 shows the approximate quantitative acute effect on blood flow of increasing the rate of metabolism in a local tissue, such as in a skeletal muscle. Note that an increase in metabolism up to eight times normal increases the blood flow acutely about fourfold.
Acute Local Blood Flow Regulation When Oxygen Availability Changes. One of the most necessary of the metabolicnutrients is oxygen. Whenever the availability of oxygen to the tissues decreases, such as (1) at high alti-tude at the top of a high mountain, (2) in pneumonia,
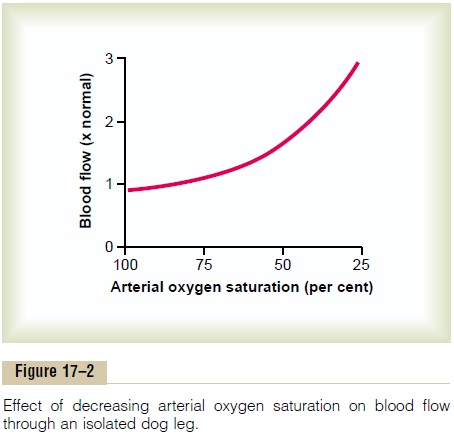
Figure 17–2 shows that as the arterial oxygen saturation decreases to about 25 per cent of normal, the blood flow through an isolated leg increases about threefold; that is, the blood flow increases almost enough, but not quite enough, to make up for the decreased amount of oxygen in the blood, thus almost maintaining an exact constant supply of oxygen to the tissues.
Total cyanide poisoning of oxygen usage by a local tissue area can cause local blood flow to increase as much as sevenfold, thus demonstrating the extreme effect of oxygen deficiency to increase blood flow.
There are two basic theories for the regulation of local blood flow when either the rate of tissue metab-olism changes or the availability of oxygen changes.
They are (1) the vasodilator theory and (2) the oxygenlack theory.
Vasodilator Theory for Acute Local Blood Flow Regu- lation—Possible Special Role of Adenosine. Accord-ing to this theory, the greater the rate of metabolism or the less the availability of oxygen or some other nutrients to a tissue, the greater the rate of formation of vasodilator substances in the tissue cells. The vasodilator substances then are believed to diffuse through the tissues to the precapillary sphincters, metarterioles, and arterioles to cause dilation. Some of the different vasodilator substances that have been suggested are adenosine, carbon dioxide, adenosinephosphate compounds, histamine, potassium ions, and hydrogen tons.
Most of the vasodilator theories assume that the vasodilator substance is released from the tissue mainly in response to oxygen deficiency.
For instance, experiments have shown that decreased availability of oxygen can cause both adenosine and lactic acid (con-taining hydrogen ions) to be released into the spaces between the tissue cells; these substances then cause intense acute vasodilation and therefore are respon-sible, or partially responsible, for the local blood flow regulation.
Many physiologists have suggested that the sub-stance adenosine is the most important of the local vasodilators for controlling local blood flow. For example, minute quantities of adenosine are released from heart muscle cells when coronary blood flow becomes too little, and this causes enough local vasodi-lation in the heart to return coronary blood flow back to normal. Also, whenever the heart becomes more active than normal and the heart’s metabolism increases an extra amount, this, too, causes increased utilization of oxygen, followed by (1) decreased oxygen concentration in the heart muscle cells with (2) consequent degradation of adenosine triphosphate (ATP), which (3) increases the release of adenosine. It is believed that much of this adenosine leaks out of the heart muscle cells to cause coronary vasodilation, pro-viding increased coronary blood flow to supply the increased nutrient demands of the active heart.
Although research evidence is less clear, many phys-iologists also have suggested that the same adenosine mechanism is the most important controller of blood flow in skeletal muscle and many other tissues as well as in the heart. The problem with the different vasodilator theories of local blood flow regulation has been the following: It has been difficult to prove that sufficient quantities of any single vasodilator sub-stance (including adenosine) are indeed formed in the tissues to cause all the measured increase in blood flow. But a combination of several different vasodila-tors could increase the blood flow sufficiently.
Oxygen Lack Theory for Local Blood Flow Control.
Although the vasodilator theory is widely accepted, several critical facts have made other physiologists favor still another theory, which can be called either the oxygen lack theory or, more accurately, the nutrient lack theory (because other nutrients besidesoxygen are involved). Oxygen (and other nutrients as well) is required as one of the metabolic nutrients to cause vascular muscle contraction. Therefore, in the absence of adequate oxygen, it is reasonable to believe that the blood vessels simply would relax and therefore naturally dilate. Also, increased utiliza-tion of oxygen in the tissues as a result of increased metabolism theoretically could decrease the availabil-ity of oxygen to the smooth muscle fibers in the local blood vessels, and this, too, would cause local vasodilation.
A mechanism by which the oxygen lack theory could operate is shown in Figure 17–3. This figure shows a tissue unit, consisting of a metarteriole with a single sidearm capillary and its surrounding tissue. At the origin of the capillary is a precapillary sphincter, and around the metarteriole are several other smooth muscle fibers. Observing such a tissue under a micro-scope—for example, in a bat’s wing—one sees that the precapillary sphincters are normally either completely open or completely closed. The number of precapillary sphincters that are open at any given time is roughly proportional to the requirements of the tissue for nutrition. The precapillary sphincters and metarteri-oles open and close cyclically several times per minute, with the duration of the open phases being propor-tional to the metabolic needs of the tissues for oxygen. The cyclical opening and closing is called vasomotion.
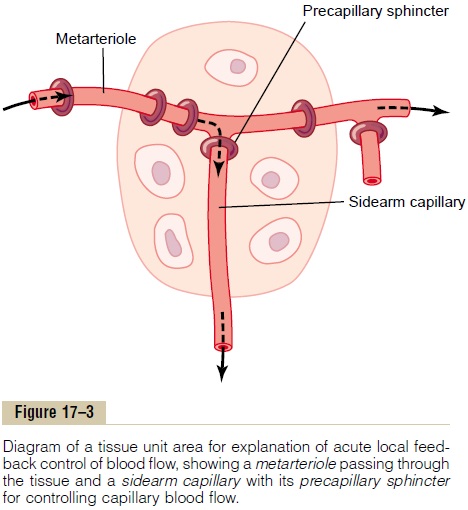
Let us explain how oxygen concentration in the local tissue could regulate blood flow through the area. Because smooth muscle requires oxygen to remain contracted, one might assume that the strength of con-traction of the sphincters would increase with an increase in oxygen concentration. Consequently, when the oxygen concentration in the tissue rises above a certain level, the precapillary and metarteriole sphincters presumably would close until the tissue cells consume the excess oxygen. But when the excess oxygen is gone and the oxygen concentration falls low enough, the sphincters would open once more to begin the cycle again.
Thus, on the basis of available data, either a vasodilator substance theory or an oxygen lack theory could explain acute local blood flow regulation in response to the metabolic needs of the tissues. Probably the truth lies in a combination of the two mechanisms.
Possible Role of Other Nutrients Besides Oxygen in Control of Local Blood Flow. Under special conditions, it has beenshown that lack of glucose in the perfusing blood can cause local tissue vasodilation. Also, it is possible that this same effect occurs when other nutrients, such as amino acids or fatty acids, are deficient, although this has not been studied adequately. In addition, vasodi-lation occurs in the vitamin deficiency disease beriberi, in which the patient has deficiencies of the vitamin B substances thiamine, niacin, and riboflavin. In this disease, the peripheral vascular blood flow everywhere in the body often increases twofold to threefold. Because these vitamins all are needed for oxygen-induced phosphorylation that is required to produce ATP in the tissue cells, one can well understand how deficiency of these vitamins might lead to diminished smooth muscle contractile ability and therefore also local vasodilation.
Special Examples of Acute “Metabolic” Control of Local Blood Flow
The mechanisms that we have described thus far for local blood flow control are called “metabolic mecha-nisms” because all of them function in response to the metabolic needs of the tissues. Two additional special examples of metabolic control of local blood flow are reactive hyperemia and active hyperemia.
Reactive Hyperemia. When the blood supply to a tissueis blocked for a few seconds to as long an hour or more and then is unblocked, blood flow through the tissue usually increases immediately to four to seven times normal; this increased flow will continue for a few seconds if the block has lasted only a few seconds but sometimes continues for as long as many hours if the blood flow has been stopped for an hour or more. This phenomenon is called reactive hyperemia.
Reactive hyperemia is another manifestation of the local “metabolic” blood flow regulation mechanism; that is, lack of flow sets into motion all of those factors that cause vasodilation. After short periods of vascu-lar occlusion, the extra blood flow during the reactive hyperemia phase lasts long enough to repay almost exactly the tissue oxygen deficit that has accrued during the period of occlusion. This mechanism emphasizes the close connection between local blood flow regulation and delivery of oxygen and other nutri-ents to the tissues.
Active Hyperemia. When any tissue becomes highlyactive, such as an exercising muscle, a gastrointestinal gland during a hypersecretory period, or even the brain during rapid mental activity, the rate of blood flow through the tissue increases. Here again, by simply applying the basic principles of local blood flow control, one can easily understand this active hyper-emia. The increase in local metabolism causes the cellsto devour tissue fluid nutrients extremely rapidly and also to release large quantities of vasodilator sub-stances. The result is to dilate the local blood vessels and, therefore, to increase local blood flow. In this way, the active tissue receives the additional nutrients required to sustain its new level of function. As pointed out earlier, active hyperemia in skeletal muscle can increase local muscle blood flow as much as 20-fold during intense exercise.
“Autoregulation” of Blood Flow When the Arterial Pressure Changes from Normal— “Metabolic” and “Myogenic” Mechanisms
In any tissue of the body, an acute increase in arterial pressure causes immediate rise in blood flow. But, within less than a minute, the blood flow in most tissues returns almost to the normal level, even though the arterial pressure is kept elevated. This return of flow toward normal is called “autoregulation of blood flow.” After autoregulation has occurred, the local blood flow in most body tissues will be related to arte-rial pressure approximately in accord with the solid “acute” curve in Figure 17–4. Note that between an arterial pressure of about 70 mm Hg and 175 mm Hg, the blood flow increases only 30 per cent even though the arterial pressure increases 150 per cent.
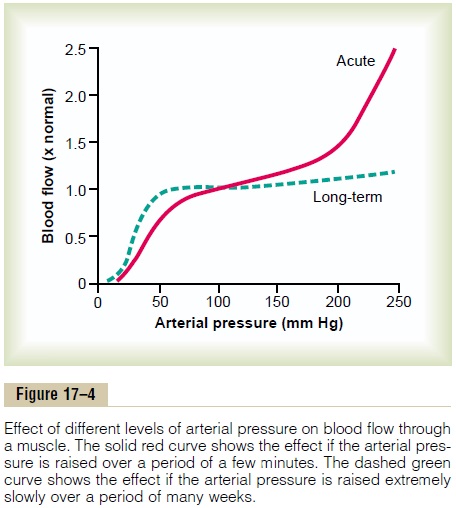
For almost a century, two views have been proposed to explain this acute autoregulation mechanism. They have been called (1) the metabolic theory and (2) the myogenic theory.
The metabolic theory can be understood easily by applying the basic principles of local blood flow regu-lation discussed in previous sections. Thus, when the arterial pressure becomes too great, the excess flow provides too much oxygen and too many other nutri-ents to the tissues. These nutrients (especially oxygen) then cause the blood vessels to constrict and the flow to return nearly to normal despite the increased pressure.
The myogenic theory, however, suggests that still another mechanism not related to tissue metabolism explains the phenomenon of autoregulation. This theory is based on the observation that sudden stretch of small blood vessels causes the smooth muscle of the vessel wall to contract for a few seconds. Therefore, it has been proposed that when high arterial pressure stretches the vessel, this in turn causes reactive vascu-lar constriction that reduces blood flow nearly back to normal. Conversely, at low pressures, the degree of stretch of the vessel is less, so that the smooth muscle relaxes and allows increased flow.
The myogenic response is inherent to vascular smooth muscle and can occur in the absence of neural or hormonal influences. It is most pronounced in arte-rioles but can also be observed in arteries, venules, veins, and even lymphatic vessels. Myogenic contrac-tion is initiated by stretch-induced vascular depolar-ization, which then rapidly increases calcium ion entry from the extracellular fluid into the cells, causing them to contract. Changes in vascular pressure may also open or close other ion channels that influence vascu-lar contraction. The precise mechanisms by which changes in pressure cause opening or closing of vas-cular ion channels are still uncertain, but likely involve mechanical effects of pressure on extracellular pro-teins that are tethered to cytoskeleton elements of the vascular wall or to the ion channels themselves.
The myogenic mechanism may be important in pre-venting excessive stretch of blood vessel when blood pressure is increased. However, the importance of the myogenic mechanism in blood flow regulation is unclear because this pressure sensing mechanism cannot directly detect changes in blood flow in the tissue. Indeed metabolic factors appear to override the myogenic mechanism in circumstances where the metabolic demands of the tissues are significantly increased, such as during vigorous muscle exercise, which can cause dramatic increases in skeletal muscle blood flow.
Special Mechanisms for Acute Blood Flow Control in Specific Tissues
Although the general mechanisms for local blood flow control discussed thus far are present in almost all tissues of the body, distinctly different mechanisms operate in a few special areas. They all are discussed throughout this text in relation to specific organs, but two notable ones are as follows:
1. In the kidneys, blood flow control is vested mainly in a mechanism called tubuloglomerular feedback, in which the composition of the fluid in the early distal tubule is detected by an epithelial structure of the distal tubule itself called the macula densa. This is located where the distal tubule lies adjacent to the afferent and efferent arterioles at the nephron juxtaglomerular apparatus. When too much fluid filters from the blood through the glomerulus into the tubular system, appropriate feedback signals from the macula densa cause constriction of the afferent arterioles, in this way reducing both renal blood flow and glomerular filtration rate back to or near to normal.
2. In the brain, in addition to control of blood flow by tissue oxygen concentration, the concentrations of carbon dioxide and hydrogen ions play very prominent roles. An increase of either or both of these dilates the cerebral vessels and allows rapid washout of the excess carbon dioxide or hydrogen ions from the brain tissues. This is important because the level of excitability of the brain itself ishighly dependent on exact control of both carbon dioxide concentration and hydrogen ion concentration.
Mechanism for Dilating Upstream Arteries When Microvascular Blood Flow Increases— The Endothelium-Derived Relaxing Factor (Nitric Oxide)
The local mechanisms for controlling tissue blood flow can dilate only the very small arteries and arterioles in each tissue because tissue cell vasodilator substances or tissue cell oxygen deficiency can reach only these vessels, not the intermediate and larger arteries back upstream. Yet, when blood flow through a micro-vascular portion of the circulation increases, this sec-ondarily entrains another mechanism that does dilate the larger arteries as well. This mechanism is the following:
The endothelial cells lining the arterioles and small arteries synthesize several substances that, when released, can affect the degree of relaxation or con-traction of the arterial wall. The most important of these is a vasodilator substance called endothelium-derived relaxing factor (EDRF), which is composedprincipally of nitric oxide, which has a half-life in the blood of only 6 seconds. Rapid flow of blood through the arteries and arterioles causes shear stress on the endothelial cells because of viscous drag of the blood against the vascular walls. This stress contorts the endothelial cells in the direction of flow and causes sig-nificant increase in the release of nitric oxide. The nitric oxide then relaxes the blood vessels. This is for-tunate because it increases the diameters of the upstream arterial blood vessels whenever microvascu-lar blood flow increases downstream. Without such a response, the effectiveness of local blood flow control would be significantly decreased because a significant part of the resistance to blood flow is in the upstream small arteries.
Related Topics