Chapter: Essentials of Psychiatry: Mood Stabilizers
Acute Bipolar Depression
Acute
Bipolar Depression
General Management Considerations
Bipolar
depression is usually indistinguishable from other forms of major depression,
except that the episodes are typically shorter and more frequent. Other clues
that the disorder may be bipolar include onset at a young age, a positive
family history for bipolar disorder (particularly in first-degree relatives),
and the occurrence of a hypomanic phase before the onset of the depressive
episode.
For the
most part, the treatment of a bipolar patient during an acute episode of
depression is similar to that of a nonbipolar patient. The same concerns about
protecting the patient from suicide and maintaining physical health and safety
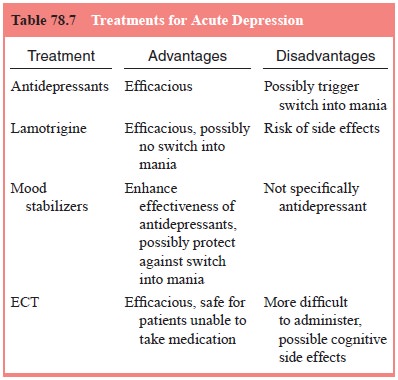
apply, and for the most part, the same antidepressants or ECT will be useful
(Table 78.7). Because of fears about speeding up cycling and/or precipitat-ing
mania in patients with bipolar disorder, most doctors avoid prescribing
tricyclics in bipolar depression. The short section that follows addresses
issues unique to treatment of bipolar depression.
Pharmacotherapy
There is
a widespread clinical impression that administering some antidepressants to
bipolar patients can trigger switches into mania. Some experts also believe
that antidepressants speed up mood cycles, although this point is more
controversial. Because of both concerns, psychiatrists are often cautious about
prescrib-ing an antidepressant at the first sign of depression in a bipolar
patient. If symptoms are mild and short-lived, many clinicians choose watchful
waiting, while maintaining close contact to de-tect clinical deterioration.
Current
guidelines by the American Psychiatric Associa-tion list lithium, as well as
the anticonvulsant lamotrigine, as first-line monotherapy for acute bipolar
depression (Hirschfeld et al., 2002).
This is a result of recent research that suggests that lamotrigine may be an effective treatment for bipolar depression,
while carrying a low risk of triggering a switch into mania.
Lamotrigine
is completely absorbed after oral administra-tion, with a bioavailability of
98%, which is not affected by food. Peak plasma concentrations occur between
1.4 and 4.8 hours after administration. Lamotrigine is approximately 55%
protein bound, making clinical interactions with other protein-bound drugs
unlikely. Its half-life is 14 to 49 hours, with steady-state levels reached in
3 to 10 days.
Lamotrigine
is metabolized predominately by glucuronic acid conjugation in the liver, with
the conjugate and the remaining 10% of the unmetabolized drug excreted in the
urine. Clearance is markedly increased with the administration of other drugs
that induce hepatic enzymes, including phenytoin, carbamazepine and
phenobarbital (Table 78.8). Adding lamotrigine to carbamazapine

can
decrease steady-state concentrations of lamotrigine by approximately 40%.
Adding lamotrigine to valproate, however, can decrease steady-state levels of
valproate by approximately 25%, while the steady-state levels of lamotrigine
increases approximately twofold. In this case, the starting dose of lamotrigine
should be lowered, and the titration made slowly.
The only
contraindication to lamotrigine use is hypersen-sitivity to the drug, though
there is a box warning about derma-tologic events, particularly rashes. These
rashes, that occurred in approximately 10% of patients, generally occur after 2
to 8 weeks and are usually macular, papular, or erythematous in nature. Of
those who develop a rash, one in 1000 adults can precede to a Stephens–Johnson
type syndrome. Because it is impossible to distinguish which rashes will
develop into this serious condition, it is advised to discontinue the
medication at any sign of drug-induced rash. Otherwise the most frequently
encountered side effects include dizziness, ataxia, somnolence, headache,
blurred vision, nausea, vomiting and diplopia.
To reduce
the risk of rash and other side effects lamotrigine should be started at low
doses and titrated slowly, especially if combined with valproate therapy.
Patients are commonly started on 25 to 50 mg p.o.q.d. doses for 2 weeks, with
doses increased by 25 to 50 mg every 1 to 2 weeks until maintenance levels
between 100 to 250 mg/day are reached. Maximum doses are lower with valproate
at about 100 to 150 mg/day. Initial labs for those un-dergoing lamotrigine
therapy should include liver function tests, renal function tests, a pregnancy
test if applicable and a complete medical work-up.
Probably
all available antidepressant drugs are effective in alleviating the symptoms of
depression in a bipolar patient, and in fact a recent 1-year retrospective
chart review by Altshuler and colleagues (2001) suggests that antidepressant
discontinuation may increase the risk of depressive relapse in bipolar
patients.
When a
patient is known to have bipolar disorder, admin-istration of an antidepressant
to reverse an acute depression is almost always used together with one of the
mood-stabilizing agents, usually lithium, valproate, carbamazepine (or a
combina-tion), or possibly one of the newer putative agents. Most bipo-lar
patients will be taking these drugs in maintenance therapy. Moreover, mood
stabilizers may enhance the effectiveness of the antidepressant and might
protect against the possibility of a switch into mania.
Nonpharmacotherapy: Electroconvulsive Therapy
ECT is a
useful alternative to antidepressants, particularly when a patient appears not
to be responding quickly or is intolerant of the medication. Early studies that
claimed that ECT works faster than medication in depressed patients are now
considered to be methodologically flawed. However, two later studies found a
more rapid decrease in depressive symptoms with ECT than is usually observed
with medications. In a total of 72 patients with major depression,
approximately six ECT treatments administered in a period of about 2 weeks led
to a decrease in mean Hamilton De-pression Rating Scale scores from between 24
and 30 to below 10 (Abrams et al.,
1991; Pettinati, 1994). The authors attributed the efficacy of treatment to the
use of high suprathreshold dosages. Sackeim and colleagues (1993) found that
higher dosage results in more rapid improvement in both bipolar and unipolar
depressed patients. There are indications that bilateral electrode placement is
superior to unilateral placement (Abrams et
al., 1991; Sackeim et al., 1993),
but there may be an increased risk of severe cogni-tive side effects with
bilateral ECT at ultrahigh dosages.
Related Topics