Chapter: Modern Pharmacology with Clinical Applications: Uterine Stimulants and Relaxants
Uterine Relaxants
UTERINE
RELAXANTS
Many risk factors are
associated with the triggering of premature labor, that is, labor that begins
before the end of week 37 of gestation. These include maternal smoking or drug
abuse, lack of prenatal care, multiple gestation, placental abnormalities,
infection of the fetal membranes, cervical incompetence, and previous preterm
birth. Although most episodes are of unknown origin, premature labor can
develop spontaneously or may follow early rupture of fetal membranes, perhaps as
a result of a genetically associated abnormality.
Uterine relaxants (tocolytic drugs) are administered where
prolonged intrauterine life would greatly benefit the fetus or would permit
additional time to allow treat-ment with drugs such as corticosteroids, which
promote the production of fetal lung surfactant. Tocolytics are also used when
temporary uterine relaxation is be de-sirable (e.g., intrauterine fetal
resuscitation). While hy-dration, bed rest, and sedation have been used to
inhibit uterine contractions, tocolytics are more likely to inhibit labor early
in gestation, especially before labor is far advanced. Agents used in this
regard include magnesium sulfate, alcohol, prostaglandin inhibitors, calcium
chan-nel blockers, hydroxyprogesterone, and β2-adrenergic agonists.
All tocolytic agents are
powerful drugs that must be used with extreme care, since pulmonary edema,
myo-cardial infarction, respiratory arrest, cardiac arrest, and death can occur
during tocolytic therapy. Newborns of mothers given tocolytics have had
respiratory depres-sion, intraventricular hemorrhage, and necrotizing
ente-rocolitis. Absolute contraindications to tocolysis include acute fetal
distress (except during intrauterine resusci-tation), chorioamnionitis,
eclampsia or severe pre-eclampsia, fetal demise (of a singleton pregnancy),
fetal maturity, and maternal hemodynamic instability.
Ethanol
Intravenous use of ethanol, while once widely employed to
inhibit premature labor, is now of historical interest only. Ethanol inhibits oxytocin release from the
pituitary and thus indirectly decreases myometrial contractility. Today, β2-adrenomimetics and magnesium
sulfate have replaced ethanol for parenteral tocolysis.
β2-Adrenoceptor Agonists
Although β2-adrenoceptor agonists are the most commonly used tocolytic agents
in the United States, they are not completely successful in treating preterm
labor. Prophylactic administration of these agents to patients at high risk for
preterm labor is not always effective. There is, however, clear evidence that β2-agonists can arrest preterm
labor for at least 48 to 72 hours. The efficacy of these drugs beyond this time
frame is in dispute. Even a short delay in delivery can be desirable, however,
in that at very early preterm gesta-tions (24–28 weeks) a 2-day delay in
delivery may mean a 10 to 15% increase in probability of survival for the
newborn. Furthermore, such a delay allows for cortico-steroid administration to
the mother, which has been shown to decrease the incidence and severity of
respi-ratory distress syndrome of the newborn, decrease the incidence of
neonatal intraventricular hemorrhage, and improve survival in the premature
newborn. Tocolysis also allows for the transport of the mother to a tertiary center
where delivery of the preterm infant often re-sults in its improved survival.
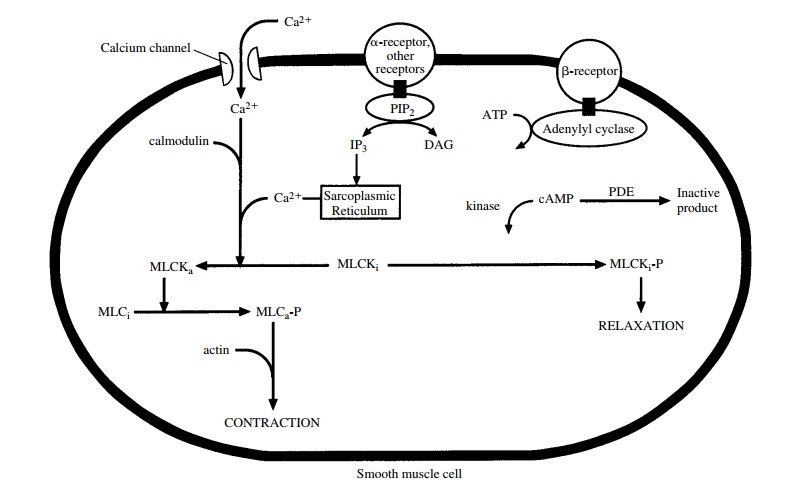
These drugs act by binding to
β2-adrenoceptors on myometrial
cell membranes and activating adenylyl cy-clase. This in turn increases levels
of cAMP in the cell (Fig. 62.1), activating cAMP-dependent protein kinase,
hence decreasing intracellular calcium concentrations and reducing the effect
of calcium on muscle contraction.
β2-Adrenergic drugs have many
side effects. These result both from their residual 1 activity and
from theirability to stimulate β2-receptors elsewhere in the body . The side effects include
palpitations, tremor, nausea, vomiting, nervousness, anxiety, chest pain,
shortness of breath, hyperglycemia, hypokalemia, and hypotension. Serious complications
of drug therapy are pulmonary edema, cardiac insufficiency, arrhyth-mias,
myocardial ischemia, and maternal death.
Terbutaline
Terbutaline (Brethine, Bricanyl) is a relatively
specific β2-adrenoceptor agonist. Terbutaline can prevent premature labor,
especially in individuals who are more than 20 weeks into gestation and have no
indication of ruptured fetal membranes or in whom labor is not far advanced.
Its effectiveness in premature labor after 33 weeks of gestation is much less
clear. Terbutaline can decrease the frequency, intensity, and duration of
uterine contractions through its ability to directly stimulate β2-adrenoceptors. While it
appears to be especially selective for β 2-receptor activation,
terbutaline does have some β 1 activity as well.
Terbutaline should be
initially used only in an appropriate hospital setting where any obstetric
com-plications can be readily addressed. After initial admin-istration, it can
be used in the outpatient setting. Concomitant use of β2-adrenergic agonists and
corti-costeroids have additional diabetic effects and may rarely lead to
pulmonary edema. The combination of β2-adrenergic agonists and magnesium sulfate can
cause cardiac disturbances, while coadministration of terbu-taline with other
sympathomimetics can lead to the po-tentiation of the actions of the latter
drugs.
Terbutaline is frequently
used in the management of premature labor, although it has not been marketed
for such use. Its effectiveness, side effects, precautions, and contraindications
are similar to those of all β2-adrener-gic agonists. Terbutaline can cause tachycardia,
hy-potension, hyperglycemia, and hypokalemia. It can be given orally in
addition to subcutaneous or intravenous administration.
Magnesium Sulfate
Magnesium sulfate prevents convulsions in preeclamp-sia and
directly uncouples excitation–contraction in myometrial cells through
inhibition of cellular action potentials. Furthermore, magnesium sulfate
decreases calcium uptake by competing for its binding sites, acti-vating
adenylyl cyclase (thereby reducing intracellular calcium), and stimulating
calcium-dependent adenosine triphosphatase (ATPase), which promotes calcium
up-take by the sarcoplasmic reticulum. Magnesium is fil-tered by the
glomerulus, so patients with low glomeru-lar filtration will have low magnesium
clearance. Although the compound does have some cardiac side effects, magnesium
sulfate may be preferred over β-adrenergic agents in patients with heart disease, dia-betes,
hypertension, or hyperthyroidism.
There is much debate as to
the efficacy of magne-sium sulfate. For effective inhibition of uterine
activity, enough must be given to maintain a blood plasma level of at least 5.5
mEq/L. Even at this level, tocolysis may be hard to achieve.
Magnesium toxicity can be
life threatening. Patients given magnesium lose patellar reflexes at plasma
levels greater than 8 to 10 mEq/L. Respiratory depression can occur at levels
greater than 10 to 12 mEq/L, with respi-ratory paralysis and arrest soon after
(e.g., at levels greater than 12–15 mEq/L). Higher levels cause cardiac arrest.
Toxicity can be avoided by following urine out-put and checking patellar
reflexes in patients receiving magnesium. Other side effects include sweating,
warmth, flushing, dry mouth, nausea, vomiting, dizzi-ness, nystagmus, headache,
palpitations, pulmonary edema, maternal tetany, profound muscular paralysis,
profound hypotension, and neonatal depression.
Other Agents
Since certain prostaglandins
are known to play a role in stimulating uterine contractions during normal
labor, it is logical that inhibitors of prostaglandin synthesis have been used
to delay preterm labor. Indomethacin (Indocin)
has been the principal agent for this use. Indomethacin is given orally or
rectally for 24 or 48 hours to delay premature labor. A potential worry
con-cerning the use of indomethacin is premature closure of the fetal ductus
arteriosus induced by its ability to inhibit prostaglandin synthesis. The fetal
ductus is more sensitive to indomethacin beyond 32 weeks of gesta-tion.
Indomethacin use also can decrease amniotic fluid volume and cause
oligohydramnios through its ability to decrease fetal urinary output. Long-term
use of ma-ternal indomethacin is associated with primary pul-monary
hypertension and an increased incidence of in-traventricular hemorrhage in the
newborn.
The calcium channel blocking
agent nifedipine, is one of the more recent drugs examined as a tocolytic
agent. It acts by impairing the entry of Ca++ into myometrial cells
via voltage-dependent channels and thereby inhibits contractility. Although
preliminary results appear promising, more studies are needed before its
usefulness can be fully as-sessed.
Hydroxyprogesterone has been used prophylacti-cally for the 12th to
37th week of pregnancy, particularly in women who are in the high-risk category
for prema-ture delivery (e.g., those with a history of premature de-livery or
spontaneous abortion). A concern relating to teratogenic potential has limited
its use. Hydroxy-progesterone as a tocolytic agent requires further eval-uation
before its routine prophylactic administration can be recommended.
With the increasing evidence
that oxytocin is impor-tant in human labor, investigators are studying oxytocin
antagonists for the treatment of preterm labor. Atosiban is an analogue of oxytocin that is modified at positions 1, 2, 4, and 8. It is a
competitive inhibitor of oxytocin binding. Early studies have demonstrated that
this drug does decrease and stop uterine contractions. Atosiban is not
available for use in the United States.
Related Topics