Chapter: Surgical Pathology Dissection : Tissue Collection for Molecular Genetic Analysis
Tissue Collection for Molecular Genetic Analysis
Tissue Collection for Molecular Genetic
Analysis
Completion
of the Human Genome Project will soon result in the identification of more than
tens of thousands of new genes. Insight into the func-tion and complex
interaction of these genes is of more than just academic interest. Indeed, an
understanding of the molecular genetic under-pinning of human disease will
fundamentally change the practice of surgical pathology.
In the
past, a major role of the prosector was to submit well-fixed tissue sections
for traditional light microscopic examination. Toward this end, the routine
handling of specimens generally involved refrigeration for variable periods of
time, fixation in formalin or other denaturing solu-tions, sampling for
microscopic evaluation, and ultimate disposal of excess tissues. The role of
the prosector is clearly evolving. These changes will first affect research
hospitals, but the pace of change is so great that soon everyone practic-ing
surgical pathology will have to be familiar with tissue collection for
molecular genetic analy-sis. In this new era of functional genomics, there is a
new emphasis on rapid collection of fresh, unfixed tissues to optimize
preservation of un-degraded DNA and RNA for genomic studies. Toward this end,
handling of specimens now emphasizes prompt dissection, avoidance of formalin
and denaturing solutions, multiplex processing for diverse diagnostic assays,
and long-term storage of excess fresh tissues. A flow diagram for the
increasingly complex and ever-evolving nature of tissue distribution is shown
in Figure 3–1.
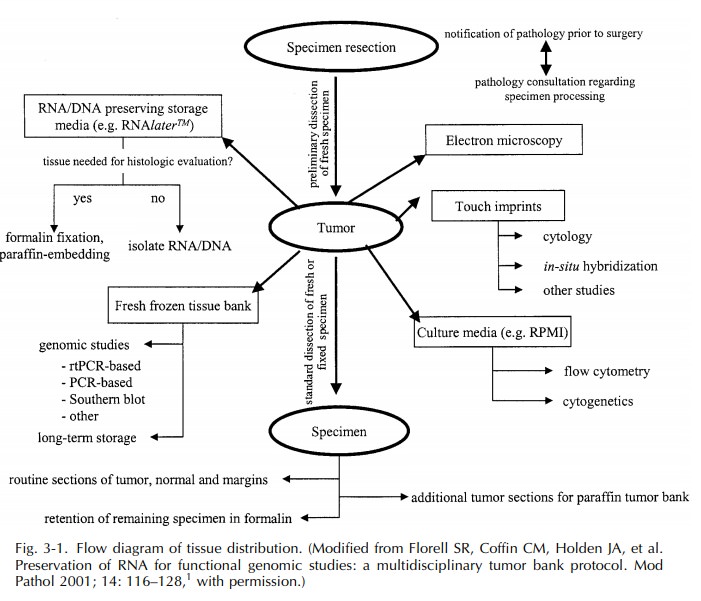
The bane
of genomic studies is the degrada-tion of RNA and DNA, and thus the major aim
when securing tissue is to do so as quickly as possible. Degradation of DNA and
RNA begins at the moment the blood supply to a tissue has been interrupted.
Therefore, rapid tissue collec-tion involves a coordinated network that begins
with punctual delivery of specimens from the operating room to the pathology
laboratory and ends with prompt processing of the specimen in the surgical
pathology suite. The time allowed from surgical resection to specimen
processing depends on a host of factors, but as a rule of thumb: the faster the better. In busy surgical
pathol-ogy laboratories, rapid tissue collection requires prioritization of
specimens potentially requiring molecular genetic evaluation over specimens
that do not. Hence, be on the lookout for hematopoi-etic tumors and primitive
tumors (i.e., “small round blue cell tumors”) of children and young adults, as
the molecular genetic profile of these tumors already plays a central role in
tumor char-acterization and patient treatment. Chromosome analysis, molecular
cytogenetics, and molecular assays are becoming increasingly useful in the
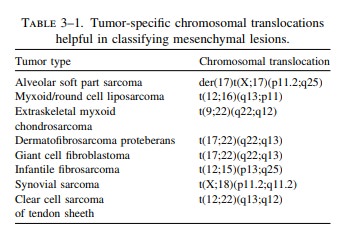
diagnosis of other tumors as well. Table 3–1 pro-vides a partial list of
heterogeneous mesenchymal lesions where the identification of certain specific
chromosomal translocations now permits more thorough and accurate
classification. If any one of these lesions is considered in the differential
diagnosis, the specimen should be targeted for rapid tissue collection.
The
optimal way to process tissues for molecu-lar genetic studies obviously depends
on the nature and methodology of the analysis. Flow cytometry, cytogenetics,
and other studies that entail the growth of living cell cultures require fresh
sterile tissue samples. These samples should be collected as 0.5- to 1.0-cm
cubes of tissue. A balanced physiologic solution such as Roswell Park Memorial
Institute (RPMI) medium serves as an excellent medium for short-term storage
and
Although various methods are being developed to optimize DNA and RNA
ex-traction from formalin-fixed tissues, polymerase chain reaction (PCR)-based
techniques looking for DNA and RNA alterations are best performed on fresh,
unfixed tissues. This tissue can be snap-frozen in liquid nitrogen and stored
for long peri-ods at 2808C. Advances are being made in the development
of more versatile tissue media (e.g., RNAlaterTM) that
preserve the integrity of DNA and RNA for molecular analysis and at the same
time maintain the histologic and immunohisto-chemical properties of the tissue.
Remember that PCR-based techniques are highly sensitive for detecting rare
abnormal cells among large num-bers of normal cells. Careful attention to
cleanli-ness, such as the use of fresh cutting utensils and changing gloves
between specimens, is therefore critical if one is to avoid the effects of
speci-men contamination.
Given
the breakneck pace at which genomic studies are finding increasing diagnostic
and therapeutic applications, it is often prudent to store excess fresh tissue
in a repository should it be needed for future analysis. This need to collect,
store, process, and distribute well-characterized human tissues for diagnostic
and investigative purposes has resulted in the emergence oftissue banks. Unlike traditional archival bankswhere the tissues
are stored as formalin-fixed and paraffin-embedded blocks, the tissues in
tissue banks are generally stored at 2808C in an unpro-cessed state or as pellets of
extracted DNA or RNA.
Not only
may these banked fresh frozen tissues be utilized for present and future
diagnostic stud-ies, they are of considerable value as resources for molecular
genetic translational research. A few guidelines should be kept in mind when
collecting and distributing tissues for investi-gative purposes. First, the
pathology laboratory should not distribute tissue for research purposes without
prior documentation of approval from the local Institutional Review Board
(IRB). IRBs have been established to define the obligations of researchers and
to ensure that the use of human tissues conform to federal regulations. Second,
patient care must always come first. There may be instances when it is simply
not possible to submit tissues for investigative studies without compromising
your ability to optimize patient care. In the case of limited specimens (e.g.,
biops-ies), there may not be enough tissue to support microscopic examination
and research studies; and for anatomically complex specimens, where it is vital
to maintain the integrity of the specimen for proper orientation and evaluation
of margins, it may not be prudent to violate the specimen to obtain fresh
tissue when formalin fixation is necessary. What ever the scenario, whenever
patient care collides with basic science require-ments, patient care must win.
Molecular
genetic analysis of human tissues is a constantly changing field. New and
exciting techniques are being developed every day. Surgi-cal pathology
prosectors familiar with the latest developments in molecular diagnoses are
best prepared to handle resected and biopsied tis-sues appropriately.
Related Topics