Chapter: SCIENCE EXPERIMENTS & AMUSEMENTS FOR CHILDREN BY CHARLES VIVIAN. Simple Technical Steped Practical Projects for school and college students.
The Power in Air Pressure
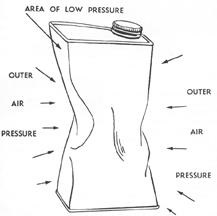
The Power in Air Pressure
You will need: Water, a screw-top can, source of
heat.
We have seen something of
air pressure at work in other experiments in this book. The present experiment
shows the quite startling pressure which air can exert under certain
circumstances.
You must obtain an empty
clean tin can with a well-fitting screw-top that makes it airtight. The larger
the can, the more spectacular the experiment will be.
Pour half a cupful of water
into the can and then place it (without the screw-top) on a stove and allow the
water to boil.
When steam is issuing from the mouth of the can, remove the can from the source of heat and screw the cap tightly into position. (Use a cloth to hold the can.)
As the can cools you will notice that its sides begin to show signs of strain. By the time the can has properly cooled, the sides will be buckled inward to such an extent that you may well be surprised that air pressure alone could have caused the damage.
When the water boiled, the steam and water vapor thus generated forced most of the air out of the can. The screw-top prevented the return of the air and as the can cooled, the steam trapped inside it condensed again into water and thus lowered the pressure inside the can.
We know that air detests vacuums or areas of low pressure and does its best to secure an entry into them. It was the pressure from the outer air as the steam inside the can condensed, which caused the sides of the can to bend inward in such a spectacular manner.
Related Topics