Chapter: Clinical Pharmacology: Anti-infective drugs
Nucleoside analogue reverse transcriptase inhibitors
Nucleoside analogue reverse transcriptase
inhibitors
NRTIs are used to treat the patient with advanced
HIV infection.
Drugs in this class include:
·
abacavir
·
didanosine
·
emtricitabine
·
lamivudine
·
stavudine
·
zidovudine.
First in the fight against AIDS
Zidovudine was the first drug to receive Food and
Drug Adminis-tration (FDA) approval for treating AIDS and AIDS-related
com-plex.
Pharmacokinetics
Each of the NRTIs has its own pharmacokinetic
properties.
Into space
Abacavir is rapidly and extensively absorbed after
oral administra-tion. It’s distributed in the extravascular space, and about
50% binds with plasma proteins. Abacavir is metabolized by the cy-tosolic
enzymes and excreted primarily in urine with the remain-der excreted in stool.
·
Lamivudine and stavudine are rapidly absorbed after adminis-tration and
are excreted by the kidneys.
·
Emtricitabine is rapidly and extensively absorbed after oral
ad-ministration and is excreted by the kidneys.
Buffer needed
Because didanosine is degraded rapidly in gastric
acid, didanosine tablets and powder contain a buffering drug to increase pH.
The exact route of metabolism isn’t fully understood. About one-half of an
absorbed dose is excreted in urine.
Well absorbed, widely distributed
Zidovudine is well absorbed from the GI tract,
widely distributed throughout the body, metabolized by the liver, and excreted
by the kidneys. The dosage may need to be adjusted in the patient with kidney
or liver disease, as is the case with most of the NRTIs.
Pharmacodynamics
NRTIs must undergo conversion to their active
metabolites to pro-duce their action.
§ Abacavir is converted to an active metabolite
that inhibits the activity of HIV-1 transcriptase by competing with a natural
compo-nent and incorporating into viral DNA.
§ Didanosine undergoes cellular enzyme
conversion to its active antiviral metabolite to block HIV replication.
§ Emtricitabine inhibits the enzyme, reverse
transcriptase, and thus inhibits viral DNA replication.
§ Lamivudine and stavudine are converted in the
cells to their ac-tive metabolites, which inhibit viral DNA replication.
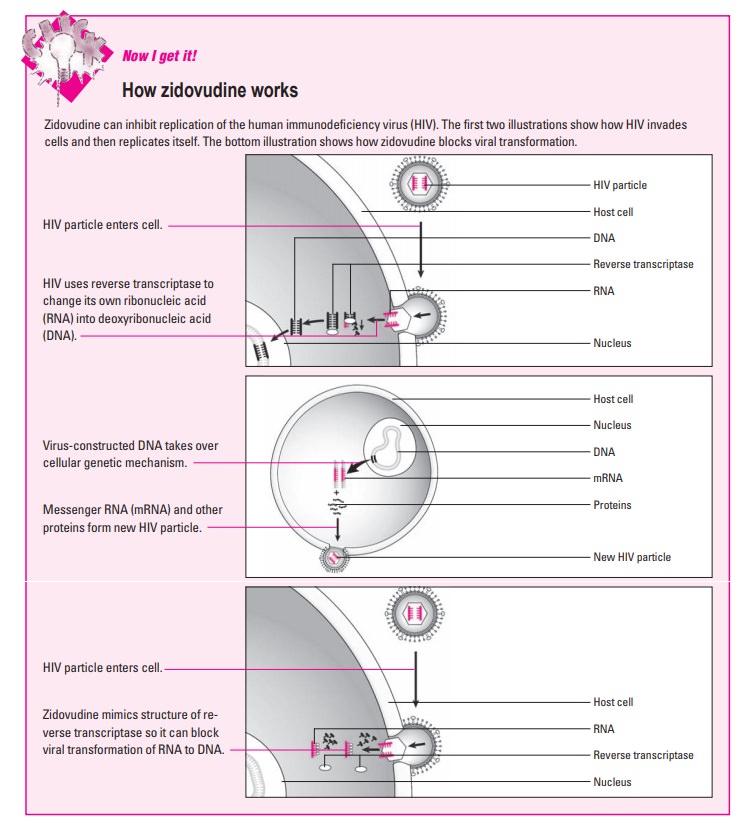
§ Zidovudine is converted by cellular enzymes
to an active form, zidovudine triphosphate, which prevents viral DNA from
replicat-ing. (See How zidovudine works)
Pharmacotherapeutics
NRTIs are used to treat HIV and AIDS.
When you’re hospitalized
I.V. zidovudine is used for the hospitalized
patient who can’t take oral medication. It’s also used to prevent transmission
of HIV from the mother to her fetus and to treat AIDS-related dementia.
Like all drugs for HIV infection, oral zidovudine
is used as part of a multidrug regimen.
Getting a jump on HIV
Didanosine, in combination with other
antiretrovirals (antivirals used to treat HIV infection), is an alternative
initial treatment for HIV infection.
Part of the combo…
Lamivudine, stavudine, and abacavir are used in
combination with other antiretrovirals to treat HIV infection. Combivir is combina-tion therapy that
includes lamivudine and zidovudine. Trizivir
is combination therapy that includes abacavir, lamivudine, and zi-dovudine; it
was approved by the FDA in November 2000 to sim-plify dosing in the treatment
of HIV.
Emtricitabine is used in combination with other
antiretrovirals to treat HIV infection.
…but be careful here
Because of inhibition of phosphorylation (the
process needed to form the active DNA-inhibiting metabolite), stavudine
shouldn’t be given in combination with zidovudine.
Drug interactions
NRTIs may be responsible for many drug
interactions.
§ Potentially fatal lactic acidosis and severe
hepatomegaly with steatosis have occurred in patients taking NRTIs alone or
with other antiretrovirals such as tenofovir. The majority of patients were
women, and obesity and prolonged NRTI exposure may be risk factors.
§ An increased risk of cellular and kidney
toxicity occurs when zi-dovudine is taken with such drugs as dapsone,
pentamidine isethionate, flucytosine, vincristine, vinblastine, doxorubicin,
in-terferon, and ganciclovir.
§ Taking zidovudine with probenecid, aspirin,
acetaminophen, in-domethacin, cimetidine, or lorazepam increases the risk of
toxici-ty of either drug.
§ Zidovudine plus acyclovir may produce
profound lethargy and drowsiness.
§ Didanosine may reduce the absorption of tetracyclines,
delavir-dine, and fluoroquinolones. Abacavir levels increase with alcohol
consumption.
§ Emtricitabine has been studied in combination
with indinavir, stavudine, famciclovir, and tenofovir; there were no clinically
sig-nificant drug interactions. (See Adverse
reactions to NRTIs.)
Related Topics