Chapter: Surgical Pathology Dissection : Laboratory Techniques
Laboratory Techniques: Decalcification
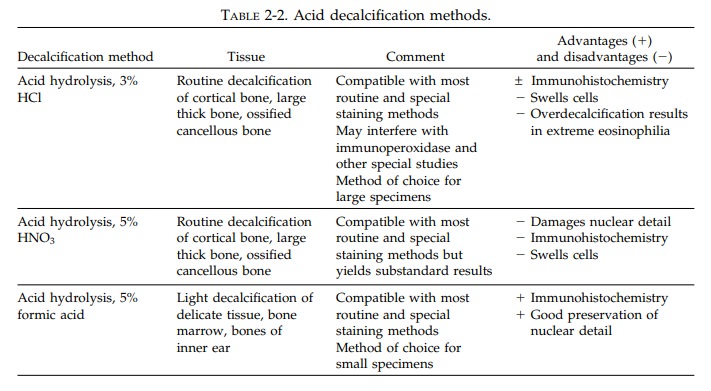
Decalcification
A wide
variety of calcified specimens are received in the surgical pathology
laboratory. While some of these should be cut with specialized, expen-sive
equipment, the vast majority of calcified specimens can be handled by a routine
histology laboratory if they are appropriately decalcified. Decalcification is
the process whereby calcium salts are removed from bone and other calcified
tissues. Three general methods are employed to decalcify tissues. These include
acid hydrolysis, organic chelation, and electrolysis (see Table 2-2). The
important points to remember about decalcifying specimens follow:
1. The
tissue must be fixed before decalcification. In most cases, fixing a specimen
for at least 24 hours in 10% neutral buffered formalin is adequate. If you use
a different fixative, make sure that the fixative employed is compati-ble with
the method of decalcification chosen.
2. Decalcification
should be carried out at room temperature and with constant magnetic stir-ring.
While heat accelerates decalcification, it also induces numerous artifacts and
thus should be avoided.
3. Do not
decalcify longer than necessary, as ex-cessive decalcification will introduce
artifacts. To avoid overdecalcification, delicate tissues should be examined
every hour and larger tissues examined as established by labora-tory protocol.
4. Residual
acid will destroy nuclear detail. Therefore, acid decalcification solutions
must be removed from bone specimens before they are processed by washing them
in water for at least 24 hours.
5. The
volume of the decalcification solution should be 10 to 15 times that of the
tissue being decalcified. These solutions should also be changed on a regular
basis.
Related Topics