Chapter: Pathology: Cellular Injury and Adaptation
Cell Death
CELL DEATH
Morphologic
types of necrosis (cell death in living tissue, often
with an inflamma-tory response) are as follows:
·
Coagulative necrosis, the most
common form of necrosis, is most often due toischemic injury (infarct). It is
caused by the denaturing of proteins within the cytoplasm. Microscopic
examination shows loss of the nucleus but preservation of cellular shape.
Coagulative necrosis is common in most organs, including the heart, liver, and
kidney, but not the brain.
·
Liquefaction necrosis results
from cellular destruction by hydrolytic enzymes,leading to autolysis (release
of proteolytic enzymes from injured cells) and het-erolysis (release of
proteolytic enzymes from inflammatory cells). Liquefaction necrosis occurs in
abscesses, brain infarcts, and pancreatic necrosis.
·
Caseous necrosis is a
combination of coagulation and liquefaction necrosis. Thegross appearance is
soft, friable, and “cheese-like.” Caseous necrosis is charac-teristic of
granulomatous diseases, including tuberculosis.
· Fat
necrosis is caused by the action of lipases on adipocytes and is
characteris-tic of acute pancreatitis. On gross examination fat necrosis has a chalky
white appearance.
· Fibrinoid
necrosis is a form of necrotic connective tissue that
histologicallyresembles fibrin. On microscopic examination fibrinoid necrosis
has an eosin-ophilic (pink) homogeneous appearance. It is often due to acute
immunologic injury (e.g., hypersensitivity type reactions II and III) and
vascular hyperten-sive damage.
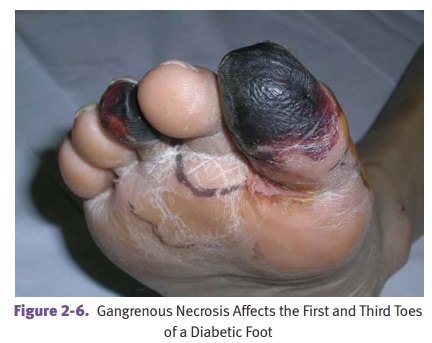
· Gangrenous necrosis is a gross term used to describe dead tissue. Commonsites of involvement include lower limbs, gallbladder, GI tract, and testes. Dry gangrene has coagulative necrosis for the microscopic pattern, while wet gan-grene has liquefactive necrosis.
Apoptosis
is a specialized form of programmed cell death without an
inflammatoryresponse. It is an active process regulated by proteins that often
affects only single cells or small groups of cells
·
In morphologic appearance, the cell shrinks in size and has dense
eosino-philic cytoplasm. Next, nuclear chromatin condensation (pyknosis) is
seen that is followed by fragmentation of the nucleus (karyorrhexis).
Cytoplasmic membrane blebs form next, leading eventually to a breakdown of the
cell into fragments (apoptotic bodies). Phagocytosis of apoptotic bodies is by
adjacent cells or macrophages.
·
Stimuli
for apoptosis include cell injury and DNA damage, lack of hor-mones,
cytokines, or growth factors, and receptor-ligand signals such as Fas binding
to the Fas ligand and tumor necrosis factor (TNF) binding to TNF receptor 1
(TNFR1).
·
Apoptosis
is regulated by proteins. The protein bcl-2 (which inhibits
apopto-sis) prevents release of cytochrome c from mitochondria and binds
pro-apop-totic protease activating factor (Apaf-1). The protein p53 (which
stimulates apoptosis) is elevated by DNA injury and arrests the cell cycle. If
DNA repair is impossible, p53 stimulates apoptosis.
·
Execution
of apoptosis is mediated by a cascade of caspases (cysteine asparticacid
proteases). The caspases digest nuclear and cytoskeletal proteins and also
activate endonucleases.
·
Physiologic
examples of apoptosis include embryogenesis
(organogenesisand development), hormone-dependent apoptosis (menstrual cycle),
thymus (selective death of lymphocytes).
·
Pathologic
examples of apoptosis include viral diseases (viral
hepatitis [Coun-cilman body]), graft-versus-host disease, and cystic fibrosis
(duct obstruction and pancreatic atrophy).
Serum
enzyme markers of cell damage include aspartate aminotransferase
(AST)(liver injury), alanine aminotransferase (ALT) (liver injury), creatine
kinase (CK-MB) (heart injury), and amylase and lipase (pancreatic injury;
amylase also rises with salivary gland injury).
Related Topics