Chapter: Plant Biology : Human uses of plants
Bioremediation
BIOREMEDIATION
Key Notes
Introduction
Bioremediation is the use of plants to extract heavy metals from contaminated soils and water. Success depends on the identification of species that can tolerate and accumulate toxins into shoots and leaves, which can then be removed and disposed of appropriately.
Hyperaccumulator
Species
Some species of plants can accumulate high levels of toxins without death. Usually, they can only tolerate a single toxic ion and grow slowly. Hyperaccumulation is thought to confer disease and pathogen resistance.
Bioremediation
Bioremediation of soils may be through the use of hyperaccumulators, genetic modification of crop species or by the use of chemical chelators. Decontamination of water can be carried out by rhizofiltration using species with high transpiration rates and by use of aquatic plants. In some instances (e.g. selenium, mercury) volatilization of the toxin to the atmosphere will also contribute to decontamination.
Introduction
Many soils are contaminated with toxic pollutants. These may be from atmospheric deposition, mine spoil, sewage sludge or contaminated ground water, or may be natural deposits of toxic ions. Some species of plant tolerate high levels of soil toxins and phytoremediation and bio-mining (the use of plants to extract minerals) has developed from the additional observation that some species accumulate toxic elements to a high level. Such species, which may accumulate more than 100 times the amounts of a toxin than other plants, are called hyperaccumulators. However, bioremediation not only depends on the use of natural hyperaccumulators, as other species may be induced to take up high levels of soil toxins either by adding chelating agents (chemicals which bind to the toxin) to the soil or by genetic modification.
Toxic ions which are found in soils and may be suitable for phytoremediation are cadmium, cobalt, copper, lead, manganese, nickel, selenium and zinc.
Hyperaccumulator species
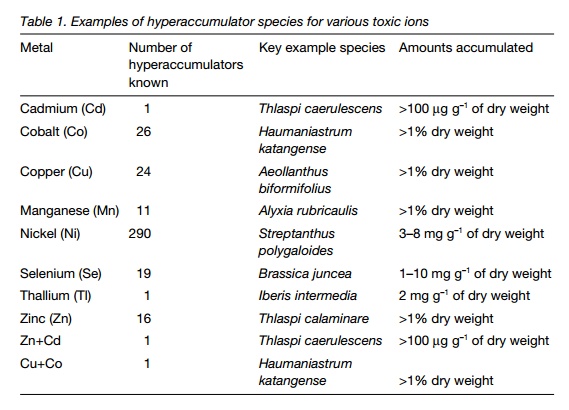
Hyperaccumulator species have been identified for most of the toxic ions found in soils, including radionuclides (Table 1). To be used successfully for bioremediation, hyperaccumulators must accumulate the toxic ion in their leaves and shoots, as removal of roots from soil is likely to be impracticable. In most instances, hyperaccumulators only grow slowly; it is assumed that the main selective advantage gained by hyperaccumulation is in deterring predators and pathogens, as tissues containing heavy metals are unpalatable and poisonous.
The fact that they only accumulate small amounts of biomass means that the total amount of an ion extracted from the soil may be small. As most hyperaccumulators can only tolerate one toxic ion, this means that they may not be effective where a soil contains several contaminants. Examples of hyperaccumulator species are shown in Table 1.
Bioremediation
Bioremediation involves the use of plants to remove toxins from soils or water. Bioremediation can be used to decontaminate soil by growing plants which accumulate the toxin and are then harvested and removed. Water can be decontaminated by rhizofiltration, in which contaminated water is passed through the roots of plants that extract the toxins and by the use of aquatic plants which are harvested and destroyed.
The species listed in Table 1 have potential for bioremediation of soils;however, because of the slow growth rates and poor yields, other alternatives are being explored. These include genetically modifying plants (e.g. arabidopsis;) and the use of chelating agents to mobilize soil toxins and reduce their toxicity to the accumulator plant. Arabidopsis has been successfully modified to express mercuric ion reductase (which converts toxic Hg2+ to Hg0which is volatilized to the atmosphere). Experiments using a chelating agent have shown that a crop of maize can accumulate >200 g of mercury per gram of shoot dry weight from contaminated soil. Volatilization to the atmosphere as a result of uptake by plants is very significant for selenium.Brassica juncea (a wild mustard) accumulates up to 10 mg g–1 (dry weight) of selenate; it also releases large quantities of dimethyl selenate to the atmosphere resulting in 25–40% loss to the air.
Several species have been indicated as useful for rhizofiltration. The best are plants with extensive root systems and which have high transpiration rates. Hybrid poplars achieved a complete removal of zinc from a solution of 800 g ml–1 in 4 h and willows (Salix sp.), Brassica juncea, sunflower (Helianthus anuus) and reeds (Phragmites) all show potential. Overall, plants like this offer a potentially effective method for the decontamination of effluent, one with negligible energy input and at low cost.
Experiments with water hyacinth (Eichhornia crassipes) show uptake of up to Cd, Co, Pb, Hg, Ni and Au from contaminated fresh water, with a total biomass production of 600 kg Ha–1 day–1. Seaweeds can be used to accumulate iodine from sea water and as indicators of local metal contamination.
Related Topics