Chapter: Clinical Anesthesiology: Anesthetic Management: Anesthesia for Patients with Neuromuscular Disease
Anesthesia for Myasthenia Gravis
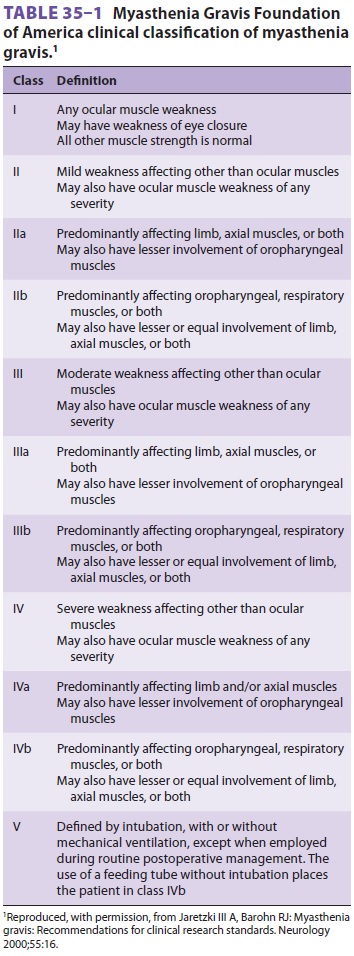
MYASTHENIA GRAVIS
Myasthenia gravis is an autoimmune disorder
char-acterized by weakness and easy fatigability of skele-tal muscle. It is
classified according to disease distribution and severity ( Table
35–1). The preva-lence is estimated at 50–200 per
million population. The incidence is highest in women during their third
decade, and men exhibit two peaks, one in the third decade and another in the
sixth decade.Weakness associated with myasthenia gravis is due to autoimmune
destruction or inactivationof postsynaptic acetylcholine receptors at the
neuro-muscular junction, leading to reduced numbers of receptors and
degradation of their function, and to complement-mediated damage to the
postsynap-tic end plate. IgG antibodies against the nicotinic acetylcholine
receptor in neuromuscular junctions are found in 85–90% of patients with
generalized myasthenia gravis and up to 50–70% of patients with ocular
myasthenia. Among patients with myas-thenia, 10–15% percent develop thymoma,
whereas approximately 70% exhibit histologic evidence of thymic lymphoid
follicular hyperplasia. Other autoimmune-related disorders (hypothyroidism,
hyperthyroidism, rheumatoid arthritis, and sys-temic lupus erythematosus) are
also present in up to 10% of patients. The differential diagnosis of
myas-thenia gravis includes a number of other clinical conditions that may
mimic its signs and symptoms
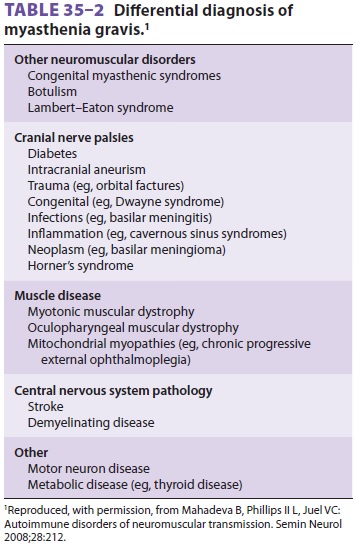
(Table 35–2). Myasthenia gravis crisis is an exacer-bation
requiring mechanical ventilation and should be suspected in any patient with
respiratory failure of unclear etiology.
The course of myasthenia gravis is marked by
exacerbations and remissions, which may be par-tial or complete. The weakness can
be asymmetric, confined to one group of muscles, or generalized. Ocular muscles
are most commonly affected, result-ing in fluctuating ptosis and diplopia. With
bulbar involvement, laryngeal and pharyngeal muscle weakness can result in
dysarthria, difficulty in chew-ing and swallowing, problems clearing
secretions, or pulmonary aspiration. Severe disease is usually also associated
with proximal muscle weakness (primar-ily in the neck and shoulders) and
involvement of respiratory muscles. Muscle strength characteristi-cally
improves with rest but deteriorates rapidly with exertion. Infection, stress,
surgery, and pregnancy
have unpredictable effects on the disease but often lead to
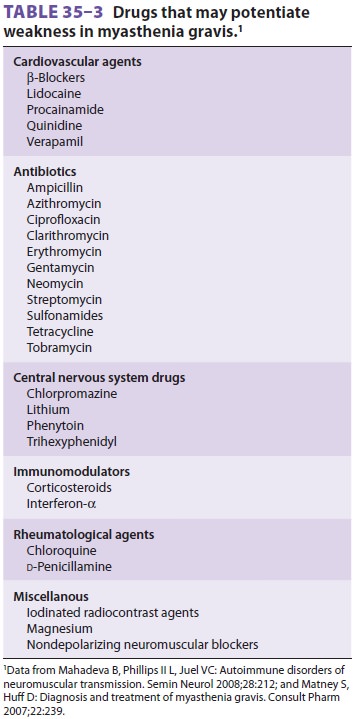
exacerbations. A number of medications may exacerbate the signs and symptoms of
myasthenia gravis ( Table 35–3).
Anticholinesterase drugs are used most
com-monly to treat the muscle weakness of this disorder. These drugs increase
the amount of acetylcholine at the neuromuscular junction through inhibition of
end plate acetylcholinesterase. Pyridostigmine is prescribed most often; when
given orally, it has an effective duration of 2–4 h. Excessive administra-tion
of an anticholinesterase may precipitate cho-linergic
crisis, which is characterized by increasedweakness and excessive
muscarinic effects, includ-ing salivation, diarrhea, miosis, and bradycardia.
An edrophonium (Tensilon) test may
help differenti-ate a cholinergic from a myasthenic crisis. Increased weakness
after administration of up to 10 mg of intravenous edrophonium indicates
cholinergic cri-sis, whereas increasing strength implies myasthenic crisis. If
this test is equivocal or if the patient clearly has manifestations of
cholinergic hyperactivity, all cholinesterase drugs should be discontinued and
the patient should be monitored in an intensive care unit or close-observation
area. Anticholinesterase drugs are often the only agents used to treat patients
with mild disease. Moderate to severe disease is treated with a combination of
an anticholinesterase drug and immunomodulating therapy. Corticosteroids are
usually tried first, followed by azathioprine, cyclo-sporine, cyclophosphamide,
mycophenolate mofetil, and intravenous immunoglobulin. Plasmapheresis is
reserved for patients with dysphagia or respira-tory failure, or to normalize
muscle strength preop-eratively in patients undergoing a surgical procedure,
including thymectomy. Up to 85% of patients younger than 55 years of age show
clinical improvement fol-lowing thymectomy even in the absence of a tumor, but
improvement may be delayed up to several years.
Anesthetic Considerations
Patients with myasthenia gravis may present
for thymectomy or for unrelated surgical or obstetric procedures, and medical
management of their con-dition should be optimized prior to the intended
procedure. Myasthenic patients with respiratory and oropharyngeal weakness
should be treated preop-eratively with intravenous immunoglobulin or
plas-mapheresis. If strength normalizes, the incidence of postoperative
respiratory complications should be similar to that of a nonmyasthenic patient
under-going a similar surgical procedure. Patients sched-uled for thymectomy
may have deteriorating muscle strength, whereas those undergoing other elective
procedures may be well controlled or in remis-sion. Adjustments in
anticholinesterase medication, immunosuppressants, or steroid therapy in the
peri-operative period may be necessary. Patients with advanced generalized
disease may deteriorate signif-icantly when anticholinesterase agents are
withheld. These medications should be restarted when the patient resumes oral
intake postoperatively. When necessary, cholinesterase inhibitors can also be
given parenterally at 301 the oral dose. Potential problems associated
with management of anticholinesterase therapy in the postoperative period
include altered patient requirements, increased vagal reflexes, and the
possibility of disrupting bowel anastomoses sec-ondary to hyperperistalsis.
Moreover, because these agents also inhibit plasma cholinesterase, they could theoretically prolong the duration of
ester-type localanesthetics and succinylcholine.
Preoperative evaluation should focus on the
recent course of the disease, the muscle groups affected, drug therapy, and
coexisting illnesses.Patients who have myasthenia gravis with respiratory
muscle or bulbar involvement areat increased risk for pulmonary aspiration.
Premedication with metoclopramide or an H2 blocker or proton pump inhibitor may decrease this risk. Because patients
with myasthenia are often very sensitive to the respiratory depressant effect
of opioids and benzodiazepines, premedication with these drugs should be done
with caution, if at all.
With the exception of NMBs, standard
anes-thetic agents may be used in patients with myasthe-nia gravis. Marked
respiratory depression, however, may be encountered following even moderate
doses of propofol or opioids. When general anesthesia is required, a volatile
agent–based anesthetic is fre-quently employed. Deep anesthesia with a volatile
agent alone in patients with myasthenia may provide sufficient relaxation for
tracheal intubation and most surgical procedures, and many clinicians
rou-tinely avoid NMBs entirely. The response to succi-nylcholine is said to be
unpredictable, but we have not found this to be so in practice. Patients may
manifest a relative resistance, or a moderately pro-longed effect . The dose of
succinyl-choline may be increased to 2 mg/kg to overcome any resistance,
expecting that the duration of paraly-sis could be increased by 5–10 min. Many
patients with myasthenia gravis are exquisitely sensitive to nondepolarizing
NMBs.Even a defasciculating dose in some patients may result in nearly complete
paralysis. If NMBs are necessary, small doses of a relatively short-acting
nondepolarizing agent are preferred. We have not found nondepolarizing NMBs to
be necessary during thymectomy with volatile anesthesia. Neuromuscular blockade
should be monitored very closely with a nerve stimulator, and ventilatory function
should be evaluated carefully prior to extubation.
Patients who have myasthenia gravis are at
risk for postoperative respiratory failure. Diseaseduration of more than 6
years, concomitant pulmo-nary disease, peak inspiratory pressure of less than −25 cm H2O (ie, −20 cm H2O), vital capacity lessthan 4 mL/kg, and
pyridostigmine dose greater than 750 mg/d are predictive of the need for
postopera-tive ventilation following thymectomy.
Women with myasthenia can experience increased weakness in the last trimester
of pregnancy and in the early postpartum period. Epidural anesthe-sia is
generally preferable for these patients because it avoids potential problems
with respiratory depression and NMBs related to general anesthesia. Excessively
high levels of motor blockade, however, can also result in hypoventilation.
Infants of myasthenic mothers may show transient myasthenia for 1–3 weeks
follow-ing birth, induced by transplacental transfer of ace-tylcholine receptor
antibodies, which may necessitate intubation and mechanical ventilation.
Related Topics