Chapter: Organic Chemistry: Alkanes and cycloalkanes
Alkanes and cycloalkanes: Drawing structures
DRAWING STRUCTURES
Key Notes
C–H Bond omission
Alkanes
can be drawn more quickly and efficiently if the C–H bonds are omitted.
Skeletal drawings
Skeletal
drawings show only the C–C bonds. Each bond junction is assumed to have a
carbon atom with sufficient hydrogens present to make up four bonds.
Alkyl groups
Alkyl
groups (CnH2n1) are alkane portions of a more
complicated structure. They can be drawn as a skeletal drawing, or as CH3,
CH2CH3, et cetera
C–H Bond omission
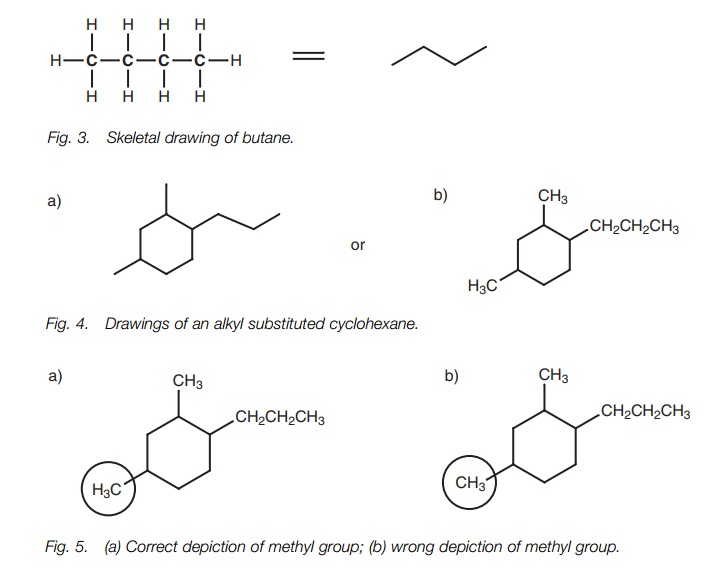
There are several ways of drawing organic
molecules. A molecule such as ethane can be drawn showing every C–C and C–H
bond. However, this becomes tedious, especially with more complex molecules,
and it is much easier to miss out the C–H bonds (Fig. 1).
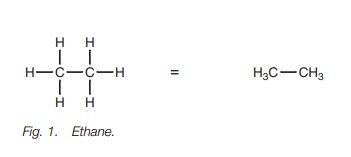
Skeletal drawings
A further simplification is often used where
only the carbon–carbon bonds are shown. This is a skeletal drawing of the
molecule (Fig. 2). With such
drawings, it is understood that a carbon atom is present at every bond junction
and that every carbon has sufficient hydrogens attached to make up four bonds.
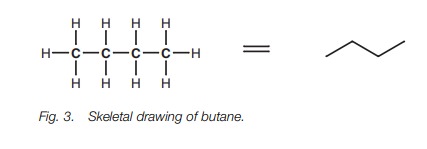
Straight chain alkanes can also be represented
by drawing the C–C bonds in a zigzag fashion (Fig. 3).
Alkyl groups
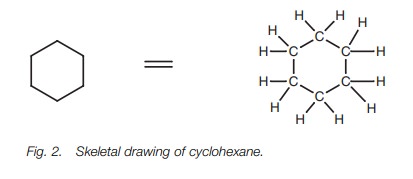
Alkyl groups (CnH2n1)
are alkane substituents of a complex molecule. Simple alkyl groups can be
indicated in skeletal form (Fig. 4a),
or as CH3, CH2CH3, CH2CH2CH3,
et cetera. (Fig. 4b).

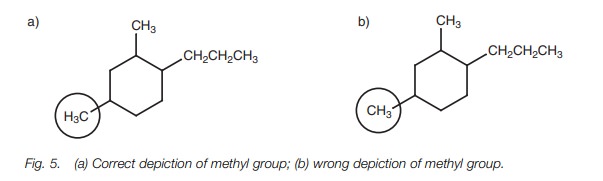
Notice how the CH3 groups have been
written in Fig. 5. The structure in Fig. 5a is more correct than the
structure in Fig. 5b since the bond
shown is between the carbons.
Related Topics