Chapter: Modern Pharmacology with Clinical Applications: Drugs Used in Neurodegenerative Disorders
ParkinsonŌĆÖs Disease
PARKINSONŌĆÖS
DISEASE
The classic publication in
1817 by James Parkinson de-fined the triad of distinguishing symptoms that bear
his name; this movement disorder is known as ParkinsonŌĆÖs disease or
parkinsonism. It generally affects the elderly and is estimated to afflict more
than 1% of individuals over the age of 65. A small subset of patients has
famil-ial forms of parkinsonism with an autosomal dominant pattern of
inheritance. Genetic mutations in three pro-teins have been identified thus
far. These genes encode for -synuclein, a protein found in abundance in
vesi-cles and synaptic regions, and for parkin and ubiquitin carboxy-terminal
hydroxylase, both of which are in-volved with protein degradation.
Some forms of parkinsonism
have been traced to specific entities, such as viral inflammation (e.g., the
pos-tencephalitic parkinsonism of the early 1900s), brain trauma, stroke, and
poisoning by manganese, carbon monoxide, pesticide, or
1-methyl-4-phenyl,-1,2,3,6-tetra-hydropyridine (MPTP). Intoxication with MPTP,
a byproduct of the synthesis of an illegal meperidine ana-logue, produces a
condition closely resembling parkin-sonism, but there is little evidence that
this or a similar toxin exists in the environment. However, the informa-tion
from research with this toxin has provided impor-tant insight into
mitochondrial function and has led to the theory that impairment of
mitochondrial function (whether of genetic or toxin derivation) may be a
rele-vant risk factor in ParkinsonŌĆÖs disease.
Although the causes of some
forms of parkinsonism are known, most cases are sporadic and are of unknown
origin (idiopathic ParkinsonŌĆÖs disease). The causes are likely multifactorial,
with genetic predisposition, envi-ronmental toxins, and aging contributing to
the initiation and progression of the disease. There is a progressive loss of
dopamine neurons with age. Relatively smooth functioning of motor control is
maintained until neu-ronal loss is such that it causes an 80% reduction of
dopamine in the striatum.At this time, clinical symptoms appear and then worsen
with increasing neuronal loss.
Another form of parkinsonism
is drug-induced, that is, iatrogenic parkinsonism, which often is a
complica-tion of antipsychotic therapy, especially following the use of the
butyrophenone and phenothiazine drug classes . Unlike idiopathic parkinson-ism,
striatal content of dopamine is not reduced by ad-ministration of these drugs.
In contrast, they produce a functional decrease in dopamine activity by
blocking the action of dopamine on postsynaptic dopamine re-ceptors.
Clinical Findings
The onset of symptoms of
ParkinsonŌĆÖs disease is usually gradual. The most prominent features of
parkinsonism are tremor, rigidity, and bradykinesia, although the time of onset
and the relative severity of each symptom may differ in individual patients.
Tremors are often unilat-eral in onset, present at rest, and cease during
voluntary movement. Rigidity, or increased muscle tone, described as jerky
resistance that has been likened to the movement of a cogwheel (cogwheel rigidity), is also an
indication of altered motor control. Bradykinesia, an extreme slowness of
movement, is the most disabling feature because it affects all motor systems.
Brady-kinesia results in a typical stooped posture when the person is standing
or walking and a characteristic shuffling gait marked by the absence of normal
arm-swinging movements. The absence of facial expression (masklike face)
results from loss of facial muscle func-tion. Inability to swallow leads to
drooling, while bradyki-nesia of the muscles in the larynx results in changes
in voice quality. Orthostatic hypotension may also be ob-served and may
complicate therapy. Cognitive dysfunc-tion and dementia are also seen in a
small percentage of ParkinsonŌĆÖs disease patients, especially the elderly.
Pathology
ParkinsonŌĆÖs disease is one of
the few neurological dis-orders in which knowledge of the pathology led
directly to the rational development of drugs to treat the dis-ease. The most prominent pathological findings in
ParkinsonŌĆÖs disease are degeneration of
the darkly pig-mented dopamine neurons in the substantia nigra, loss of
dopamine in the neostriatum, and the presence of intra-cellular inclusion
bodies known as Lewy bodies. Other neuronal
populations are also affected in ParkinsonŌĆÖs disease to a much lesser extent,
but they may contribute to some of the other pathology seen in parkinsonism
(e.g., cognitive decline, depression, and dementia).
In postmortem examination of
tissue, the substantia nigra is readily identifiable because of the dark pigmen-tation
in the neurons that is the result of the accumula-tion of neuromelanin, a
substance whose neurochemical composition is not completely known but is
thought to derive from oxidized dopamine. Lewy bodies are com-posed of many
cytoskeleton and other proteins, includ-ing -synuclein, ubiquitin, and
synaptophysin. It is not clear whether the formation of these inclusions
con-tributes to neuronal degeneration or they are merely a byproduct of
degenerating neurons.
Basal Ganglia Anatomy
The basal ganglia can be
viewed as modulators of mo-tor function. They are composed of several brain
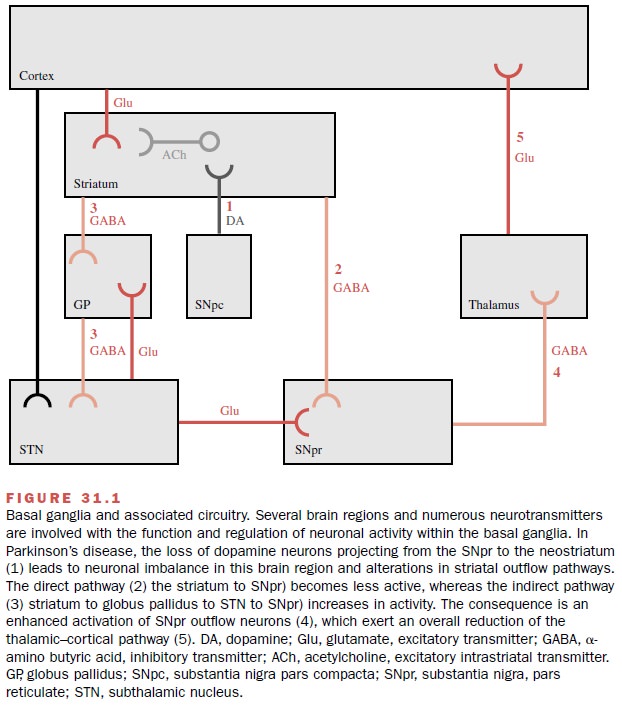
re-gions, including the neostriatum and the substantia ni-gra (Fig. 31.1). The
neostriatum receives massive excitatory input from the cortex that is mediated
by neurons that use glutamate as the neurotransmitter. The dopamine neurons
originate in the substantia nigra pars
compacta and project to the
neostriatum, where they synapse on
the input glutamatergic terminals and on striatal projection neurons that use
the neurotransmit-ter ╬│-Aminobutyric acid (GABA).
Dopamine is a catecholamine
whose actions are mediated by dopamine receptors that are classified as D1-like
(D1, D5) or D2-like (D2, D3,
D4). Dopamine actions on D1 receptors ex-ert an
excitatory effect, whereas the actions of dopamine on D2 receptors
inhibit neuronal activity. The loss of striatal dopamine produces an imbalance
in in-formation processing in the neostriatum that modifies transmission in
other basal ganglia regions. Also impor-tant in neural transmission are the
striatal interneurons that are found within the confines of the striatum, that
use the excitatory neurotransmitter acetylcholine, and that modulate the
activity of striatal output neurons.
Possible Mechanisms of Neurodegeneration
The mechanisms responsible
for the degeneration of dopamine neurons are not known, but hypotheses in-clude
effects such as oxidative stress and excitotoxicity.
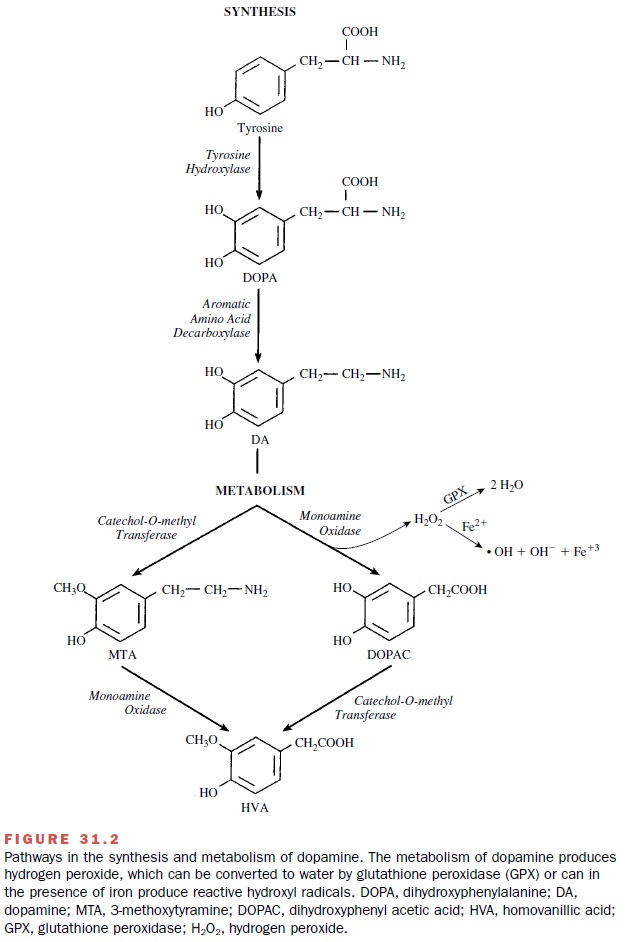
The inability of the neurons
to eliminate the oxidative load may result in a self-perpetuating cycle of
oxidative damage that ultimately leads to neuronal death. One source of
oxidative stress may be dopamine metabolism (Fig. 31.2). The excessive
excitatory activity in the sub-stantia nigra created by the loss of dopamine
actions within the striatum could lead to excitotoxicity that is mediated by
glutamate.
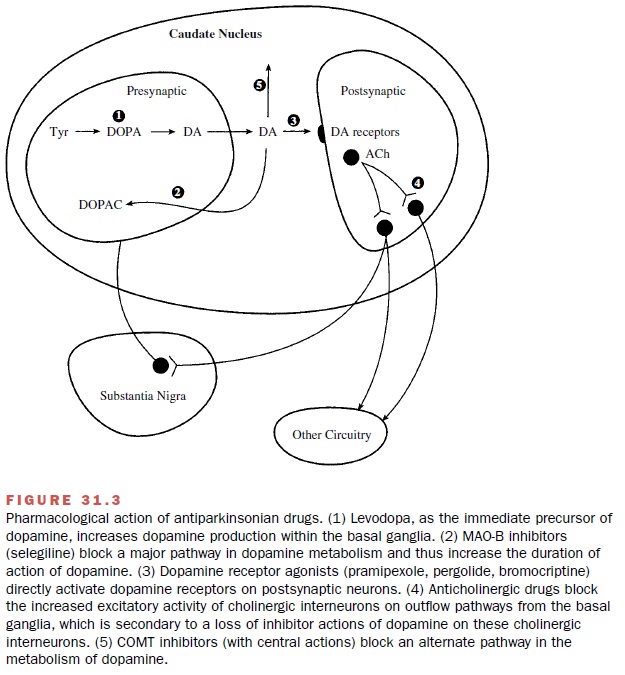
Therapy of Parkinsonism
Since there is no cure for
parkinsonism, the aim of phar-macological therapy is to provide symptomatic
relief. This is obtained through the use of drugs that either in-crease dopaminergic
actions or diminish neuronal out-flow from the striatum. These drugs include
levodopa, which increases brain dopamine levels; dopamine ago-nists, which
directly stimulate dopamine receptors; monoamine oxidase (MAO) inhibitors,
which prevent dopamine metabolism; and anticholinergic agents, which reduce the
excitatory activity within the striatum (Fig 31.3).
Related Topics