Chapter: Health Management in Aquaculture: Physical, environmental, and chemical methods of disease prevention and control
Chemical methods of disease prevention and control in Aquaculture
CHEMICAL METHODS
Prophylactic Methods
Prophylactic treatment methods are protective or defensive measures designedto prevent a disease from occurring. They are used to combat external parasites and stress-mediated bacterial diseases.
Disinfecting culture facilities
Tanks – Rearing tanks should be disinfected in between rearing periods. Drainand scrub tank bottom and sidewalls using powdered detergent and plastic brush to remove debris. Rinse thoroughly to remove soap suds and loosened contaminants. Disinfect with 200-ppm chlorine for 1 h or with 100-ppm chlorine for several hours. Scrub tank bottom and sidewalls again. Rinse several times with clean freshwater and dry under the sun.
Earthen ponds – Drain the pond and then dry. Apply lime (0.5-1 ton/ha CaCO3or agricultural lime) and 20-ppm tea seed cake, or any of 600-ppm Roccal (ben-zalkonium chloride), Hyamine 1622 and Hyamine 3500 (quaternary com-pounds).
Disinfecting rearing water
Chlorination method — Chlorine is the cheapest disinfectant. One of the bestand commonly used is calcium hypochlorite (powder form) or ordinary house-hold bleach (Purex, Chlorox). Filter the water first. Chlorine loses its strength when exposed to air. It is reduced by organic matter (mud, slime, plant matter) and must be covered. Use 5 to 20-ppm chlorine for 12-24 h, then neutralize with sodium thiosulfate until residual chlorine becomes zero. Chlorinated, neutralized water must be used within 6 h as bacterial load increases after 12 h.
Ozonation method – Ozone (O3-triatomic oxygen) is a more powerful oxidizingagent than hypochlorite. It can de-activate or destroy viruses and bacteria that might be transmitted through the water supply system. At 90-mg/L concentra-tion and exposure for 20 min, ozone can control bacterial and viral fish patho-gens in water supplies, although this level may not eliminate 100% of patho-gens. Like chlorine, ozone is toxic to aquatic organisms. Oxygen (O2) is a breakdown product of ozone, and oxidizing action may result in oxygen super-saturation or gas-bubble disease. The concentration of 0.005-ppm O3 is the up-per limit for continuous exposure. Ozonated water must be re-aerated before it is used.
Disinfecting materials
Materials like pails, brushes, scoop nets, secchi disk, glasswares, hose, etc. may be disinfected in between use in different culture facilities. Dip the materials in 400-ppm chlorine solution for a few seconds, and rinse thoroughly with clean water.
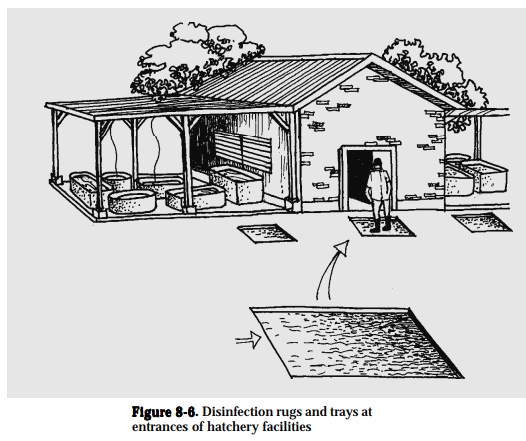
Disinfect footwear by placing 200-ppm chlorine or 3% Lysol solution in disin-fecting rugs/trays at the entrance of aquaculture facility (Fig. 8-6). Wash rugs and change disinfectant regularly.
Disinfecting feeds
Artemia cysts – Cysts may be decapsulated in chlorine solution. Use 30 ppmchlorine or 10 ppm formalin, 1 h before hatching.
Disinfecting the hosts (especially Penaeus monodon)
Spawners – Disinfect with 5 ppm Treflan for 1 h or 50-100 ppm formalin for 30-60 min. Rinse spawners thoroughly in clean water.
Eggs – Disinfect with 20 ppm detergent for 2-4 h. Disinfection should be done at least 6 h before hatching. Rinse thoroughly and completely change water in hatching tank.
Larvae – Disinfect with 0.1 ppm Treflan (trifluralin) once every other day.
Chemotherapy
Chemotherapy involves the use of drugs or chemicals for treating infectious diseases. It is considered as the method of “last resort” in any disease control program.
Factors to consider before using chemicals:
Tolerance of the host to the chemical – Tolerance of fish varies with age, size, species, and health condition. Younger or smaller fish are more sensi-tive than bigger or older ones. Some species are better able to tolerate chemicals than others are. Fish weakened by disease become less tolerant to stress and environmental fluctuations.
• Efficiency of the chemical – The choice of what chemical to use is based on differential toxicity, that is, the chemical must be lethal to the target micro-organism but harmless to the host. It is essential to know the properties of the chemical such as the active ingredient, solubility and application method. The chemical must not harm the environment.
• Restrictions on the use of chemicals to treat food fish – Use only chemicals that break down rapidly and are eliminated quickly from all fish tissues to avoid tissue residue problems. The chemical must not form toxic or carcino-genic products during cooking of the contaminated flesh.
• Consequences of drug resistance – The indiscriminate use of antibiotics may lead to the development of drug-resistant strains.
• Economics – Chemicals are expensive, and one should know the value of the stock and the cost of treatment to determine the benefits that may be derived from their use.
The methods of chemical treatment are as follows:
1. External methods – These are used to control ectoparasites and other micro-organisms outside the fish. They are employed to reduce or eliminate potential pathogens from tanks, ponds, and from other materials. The external method may either be topical or by immersion.
Topical
This is the direct and simplest method for treating wounds, skin ulcers and other localized infection. Immobilize the fish before taking it out of the wa-ter for treatment. Apply the drug directly on the infected area. The method is labor-intensive and should be used only for high-value fish.
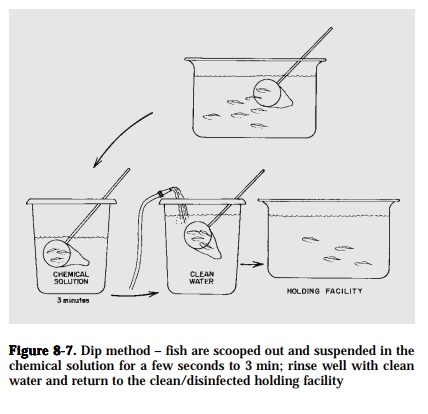
Immersion
Dip. Place the fish in a scoop net and immerse in a high concentration ofchemical solution for a specified time, usually from a few seconds to a few minutes. Rinse the fish immediately in clean water after treatment and return the fish to the clean/disinfected holding facility (Fig. 8-7).
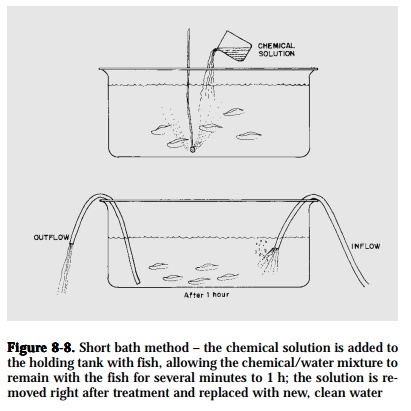
Short bath.Add the chemical solution to the holding facility where the fishare to be treated, allowing the fish to remain in the chemical and water mixture for a designated time, usually a few hours or less (Fig. 8-8). After treatment, remove treated water immediately and replace with clean water.
Flush. Add a highly concentrated chemical solution at the water inlet andallow this to pass through the water flow system and out of the effluent pipe (Fig. 8-9).
Long bath. Treat the fish for a longer time, usually 12 h or more, in a chemi-cal solution of low concentration.
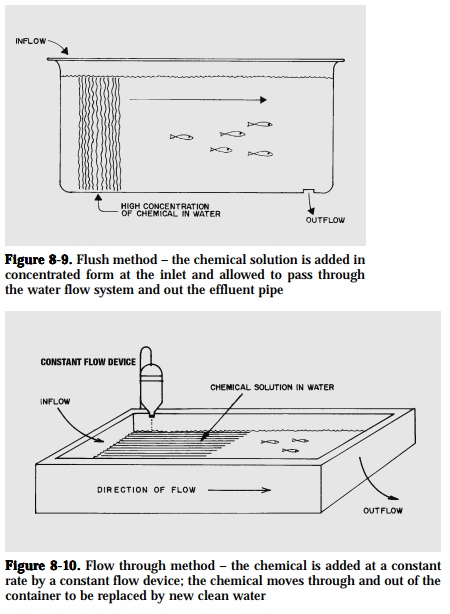
Flow-through. Add the chemical at a constant rate through a meteringdevice to give a consistent low concentration for the desired treatment time. The treated water moves through and out of the holding facility, and is replaced by new clean water (Fig. 8-10).
2. Systemic treatment – This is employed for treatment of systemic infections.Chemicals are added into the feed. The advantages of this method are that fewer chemicals are needed, environmental pollution is lessened and labor in-put is low. The disadvantages are the non-feeding of sick fish and that, since some drugs are not stable in moist diets, this would require introduction of more palatable components.
3. Parenteral treatment – This is the direct and most effective route of drugadministration. Advantages are that accurate dosage can be administered and pollution of the environment is avoided. The disadvantages are that it is labor-intensive, it contributes to handling stress and it is good only for big and valu-able stocks.
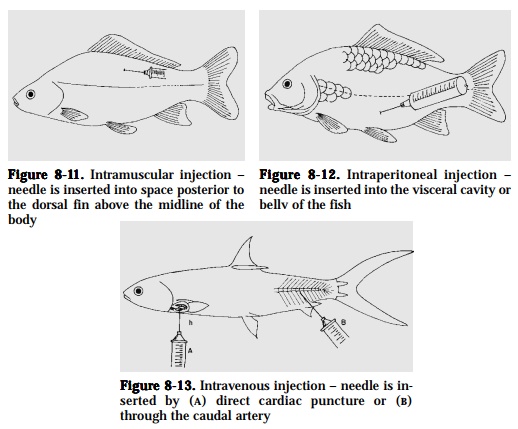
Intramuscular.Insert the needle posterior to the dorsal fin above the midline ofthe body (Fig. 8-11). Absorption is slow (not very effective) or, sometimes, does not take place at all.
Intraperitoneal. This is the most common method of injection. Insert theneedle into the visceral cavity or belly of the fish (Fig. 8-12). The drug must be highly absorbable and should be able to pass through either the intesti-nal wall or some other membrane to be absorbed into the fish system.
Intravenous.Insert needle by direct cardiac puncture, or through the caudalartery (Fig. 8-13). This results in rapid dispersal and is the most effectiveroute for administering antibiotics. The only drawback is that this can beused only on large fish.
Related Topics