Chapter: Organic Chemistry: Aldehydes and ketones
Properties of Aldehydes and ketones
PROPERTIES
Key Notes
Carbonyl group
The
carbonyl group is a C=O group. The carbonyl group is planar with bond
angles of 120┬░, and
consists of two sp2
hybridized atoms (C and O) linked by a strong Žā bond and a weaker ŽĆ bond. The carbonyl group is polarized such
that oxygen is slightly negative and carbon is slightly posi-tive. In aldehydes
and ketones, the substituents must be one or more of the following ŌĆō an alkyl
group, an aromatic ring, or a hydrogen.
Properties
Aldehydes
and ketones have higher boiling points than alkanes of compa-rable molecular
weight due to the polarity of the carbonyl group. However, they have lower
boiling points than comparable alcohols or carboxylic acids due to the absence
of hydrogen bonding. Aldehydes and ketones of small molecular weight are
soluble in aqueous solution since they can participate in intermolecular
hydrogen bonding with water. Higher molecular weight aldehydes and ketones are
not soluble in water since the hydrophobic char-acter of the alkyl chains or
aromatic rings outweighs the polar character of the carbonyl group.
Nucleophilic and electrophilic centers
The
oxygen of the carbonyl group is a nucleophilic center. The carbonyl car-bon is
an electrophilic center.
KetoŌĆōenol tautomerism
Ketones
are in rapid equilibrium with an isomeric structure called an enol. The keto
and enol forms are called tautomers and the process by which they interconvert
is called ketoŌĆōenol tautomerism. The mechanism can be acid or base catalyzed.
Spectroscopic analysis of aldehydes and ketones
Aldehydes
and ketones show strong carbonyl stretching absorptions in their IR spectra as
well as a quaternary carbonyl carbon signal in their 13C nmr
spectra. Aldehydes also show characteristic CŌĆōH stretching absorp-tions in
their IR spectra and a signal for the aldehyde proton in the 1H nmr
which occurs at high chemical shift. The mass spectra of aldehydes and ketones
usually show fragmentation ions resulting from cleavage next to the carbonyl
group. The position of the uv absorption band is useful in the structure
determination of conjugated aldehydes and ketones.
Carbonyl group
Both aldehydes and ketones contain a carbonyl
group (C=O). The substituents attached to the carbonyl
group determine whether it is an aldehyde or a ketone, and whether it is
aliphatic or aromatic.
The geometry of the carbonyl group is planar
with bond angles of 120┬░. The carbon and oxygen atoms of the carbonyl
group are sp2 hybridized
and the double bond between the atoms is made up of a strong Žā bond and a weaker ŽĆ bond. The carbonyl bond is shorter than a CŌłÆO single bond (1.22 ├ģ vs. 1.43 ├ģ) and is also stronger since two
bonds are present as opposed to one (732 kJ molŌłÆ1 vs. 385 kJ molŌłÆ1). The carbonyl group is more reactive than a CŌłÆO single bond due to the relatively weak ŽĆ bond.
The carbonyl group is polarized such that the
oxygen is slightly negative and the carbon is slightly positive. Both the
polarity of the carbonyl group and the presence of the weak ŽĆ bond explains much of the chemistry and the physical properties of
aldehydes and ketones. The polarity of the bond also means that the carbonyl
group has a dipole moment.

Properties
Due to the polar nature of the carbonyl group,
aldehydes and ketones have higher boiling
points than alkanes
of similar molecular
weight. However, hydrogen bonding is not possible between
carbonyl groups and so aldehydes and ketones have lower boiling points than
alcohols or carboxylic acids.
Low molecular weight aldehydes and ketones
(e.g. formaldehyde and acetone) are soluble in water. This is because the
oxygen of the carbonyl group can partic- ipate in intermolecular hydrogen
bonding with water molecules. As molecular weight increases, the hydrophobic character
of the attached alkyl chains starts to outweigh the water solubility of the
carbonyl group with the result that large molecular weight aldehydes and
ketones are insoluble in water. Aro-matic ketones and aldehydes are insoluble
in water due to the hydrophobic aro- matic ring.
Nucleophilic and electrophilic centers
Due to the polarity of the carbonyl group,
aldehydes and ketones have a nucleophilic
oxygen center and an electrophilic
carbon center as
shown for propanal. Therefore,
nucleophiles react with aldehydes and ketones at the carbon center, and
electrophiles react at the oxygen center.
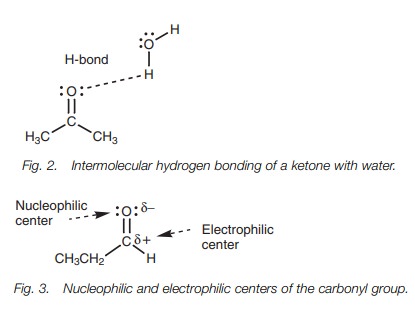
KetoŌĆōenol tautomerism
Ketones which have hydrogen atoms on their ╬▒-carbon (the carbon next to the carbonyl group) are in
rapid equilibrium with an isomeric structure called an enol where the
╬▒-hydrogen ends up on the oxygen instead of the carbon. The two isomeric forms
are called tautomers and the process of
equilibration is called tautomerism (Fig. 4). In general, the equilibrium
greatly favors the keto tautomer and the enol tautomer may only be present in
very small quantities.
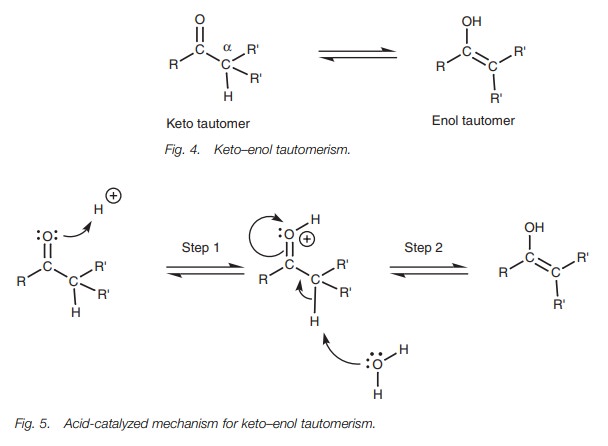
The tautomerism mechanism is catalyzed by acid
or base. When catalyzed byacid (Fig. 5), the carbonyl group acts as a
nucleophile with the oxygen using a lone pair of electrons to form a bond to a
proton. This results in the carbonyl oxygen gaining a positive charge which
activates the carbonyl group to attack by weak nucleophiles (Step 1). The weak
nucleophile in question is a water molecule which removes the ╬▒-proton from the
ketone, resulting in the formation of a new C = C double bond and cleavage of
the carbonyl ŽĆ bond. The enol tautomer is formed thus neutralizing the
unfavorable positive charge on the oxygen (Step 2).
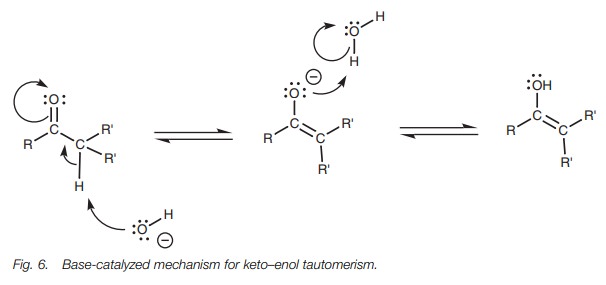
Under basic conditions (Fig. 6), an enolate ion
is formed, which then reacts with water to form the enol.
Spectroscopic analysis of aldehydes and ketones
The IR spectra of aldehydes and ketones are characterized by strong absorptions due to C=O stretching. These occur in the region 1740ŌĆō1720 cmŌłÆ1 for aliphatic aldehydes and 1725ŌĆō1705 cmŌłÆ1 for aliphatic ketones. However conjugation to aromatic rings or alkenes weakens the carbonyl bond resulting in absorptions at lower wavenumbers.
For example, the carbonyl
absorptions for aromatic aldehydes and ketones are in the regions 1715ŌĆō1695 cmŌłÆ1 and 1700ŌĆō1680 cmŌłÆ1 respectively. For cyclic ketones, the
absorption shifts to higher wavenumber with increasing ring strain. For
example, the absorptions for cyclohexanone and cyclobutanone are 1715 and 1785
cmŌłÆ1 respectively.
In the case of an aldehyde, two weak
absorptions due to CŌĆōH stretching of the aldehyde proton may be spotted, one in
the region 2900ŌĆō2700 cmŌłÆ1 and the other close to 2720 cmŌłÆ1. The aldehyde proton gives a characteristic
signal in the 1H nmr in the region 9.4ŌĆō10.5 ppm. If the aldehyde
group is linked to a carbon bearing a hydrogen, coupling will take place,
typically with a small coupling constant of about 3 Hz. Indications of an
aldehyde or ketone can be obtained indirectly from the 1H nmr by the
chemical shifts of neighboring groups. For example, the methyl signal of a
methyl ketone appears at 2.2 ppm as a singlet.
The carbonyl carbon can be spotted as a
quaternary signal in the 13C nmr spec-trum in the region 200ŌĆō205 ppm
for aliphatic aldehydes and 205ŌĆō218 ppm for aliphatic ketones. The
corresponding regions for aromatic aldehydes and ketones are 190ŌĆō194 ppm and
196ŌĆō199 respectively.
The mass spectra of aldehydes and ketones often
show fragmentation ions resulting from bond cleavage on either side of the
carbonyl group (╬▒-cleavage). Aromatic aldehydes and ketones
generally fragment to give a strong peak at m/e 105 due to the benzoyl
fragmentation ion [PhC=O]+.
The carbonyl groups of saturated aldehydes and
ketones give a weak absorp-tion band in their uv spectra between 270 and 300
nm. This band is shifted to longer wavelengths (300ŌĆō350 nm) when the carbonyl
group is conjugated with a double bond. The exact position of the uv absorption
band can be useful in the structure determination of conjugated aldehydes and
ketones.
Related Topics