Chapter: Clinical Anesthesiology: Anesthetic Equipment & Monitors : Cardiovascular Monitoring
Invasive Arterial Blood Pressure Monitoring
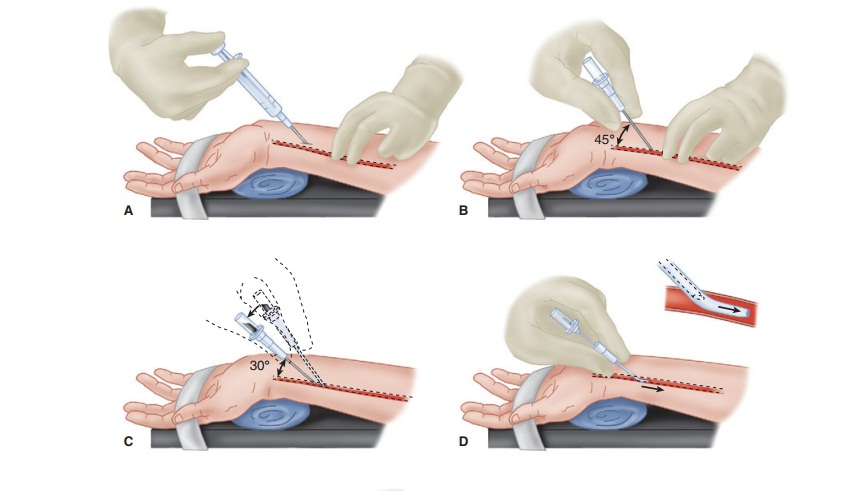
Invasive Arterial Blood Pressure Monitoring
Indications for invasive arterial blood
pressure monitoring by catheterization of an artery include induced current or
anticipated hypotension or wide blood pressure deviations, end-organ disease
neces-sitating precise beat-to-beat blood pressure regula-tion, and the need
for multiple arterial blood gas measurements.
Contraindications
If possible, catheterization should be
avoided in smaller end arteries with inadequate collateral blood flow or in
extremities where there is a suspicion of preexisting vascular insufficiency.
A. Selection of Artery for Cannulation
Several arteries are available for
percutaneous catheterization.
The radial
artery is commonly cannulated because of its superficial location and
substantial collateral flow (in most patients the ulnar artery is larger than
the radial and there are connections between the two via the palmar arches).
Five percent of patients have incomplete palmar arches and lack adequate collateral
blood flow. Allen’s test is a simple, but not reliable, method for assessing
the safety of radial artery cannulation. In this test, the patient
exsanguinates his or her hand by making a fist. While the operator occludes the
radial and ulnar arteries with fingertip pressure, the patient relaxes the
blanched hand. Collateral flow through the palmar arterial arch is confirmed by
flushing of the thumb within 5 sec after pressure on the ulnar artery is
released. Delayed return of normal color (5–10 s) indicates an equivocal test
or insufficient collateral circulation (>10 s). The Allen’s test is of such questionable
utility that many practitioners routinely avoid it. Alternatively, blood flow
distal to the radial artery occlusion can be detected by palpation, Doppler
probe, plethysmography, or pulse oximetry. Unlike Allen’s test, these methods
of determining the adequacy of collateral circulation do not require patient
cooperation.
Ulnar
artery catheterization
is usually moredifficult than radial catheterization because of the ulnar
artery’s deeper and more tortuous course. Because of the risk of compromising
blood flow to the hand, ulnar catheterization would not normally be considered
if the ipsilateral radial artery has been punctured but unsuccessfully
cannulated.
The brachial
artery is large and easily identifiable in the antecubital fossa. Its
proximity to the aorta provides less waveform distortion. However, being near
the elbow predisposes brachial artery catheters to kinking.
The femoral
artery is prone to atheroma formation and pseudoaneurysm, but often
provides an excellent access. The femoral site has been associated with an
increased incidence of infectious complications and arterial thrombosis.
Aseptic necrosis of the head of the femur is a rare, but tragic, complication
of femoral artery cannulation in children.
The dorsalis
pedis and posterior tibialarteries are some distance from the aorta
andtherefore have the most distorted waveforms.
The axillary
artery is surrounded by the axillary plexus, and nerve damage can result
from a hematoma or traumatic cannulation. Air or thrombi can quickly gain
access to the cerebral circulation during vigorous retrograde flushing of
axillary artery catheters.
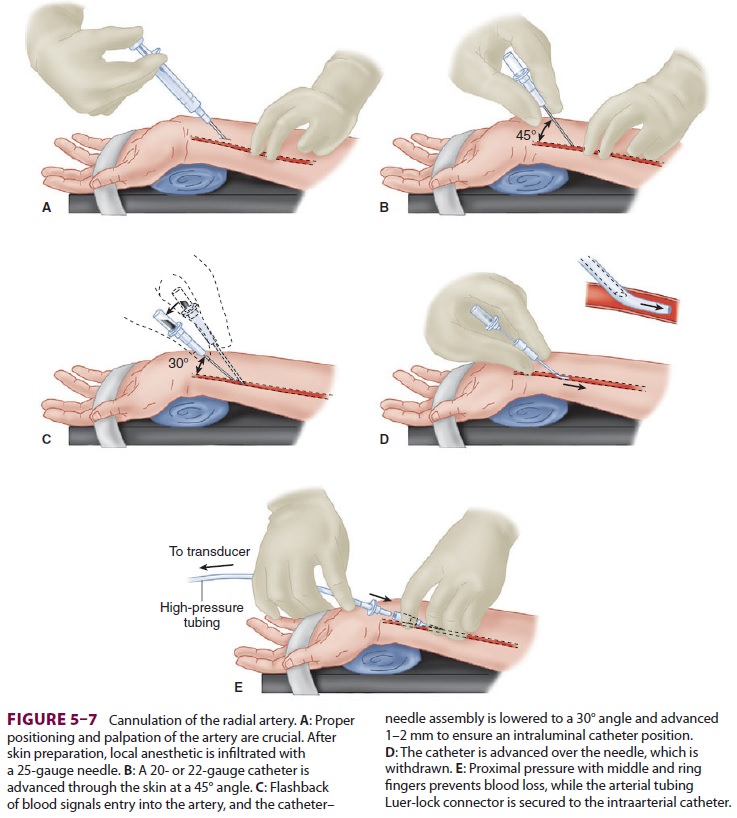
B. Technique of Radial Artery Cannulation
One technique of radial artery cannulation is
illus-trated in Figure
5–7. Supination and extension of the wrist provide optimal exposure
of the radial artery. The pressure–tubing–transducer system should be nearby
and already flushed with saline to ensure easy and quick connection after
cannula-tion. The radial pulse is palpated, and the artery’s course is
determined by lightly pressing the tips
of the index and middle fingers of the anesthesiolo-gist’s nondominant hand
over the area of maximal impulse or by use of ultrasound. Using aseptic
tech-nique, 1% lidocaine is infiltrated in the skin of awake patients, directly
above the artery, with a small gauge needle. A larger gauge needle can then be
used as a skin punch, facilitating entry of an 18-, 20-, or 22-gauge catheter
over a needle through the skin at a 45° angle, directing it toward the point of
palpation. Upon blood flashback, a guidewire may be advanced through the
catheter into the artery and the cath-eter advanced over the guidewire.
Alternatively, the needle is lowered to a 30° angle and advanced another 1–2 mm
to make certain that the tip of the catheter is well into the vessel lumen. The
catheter is advanced off the needle into the arterial lumen, after which the
needle is withdrawn. Applying firm pressure over the artery proximal to the
catheter tip, with the middle and ring fingertips, prevents blood from spurting
from the catheter while the tubing is connected. Waterproof tape or suture can
be used to hold the catheter in place.
C. Complications
Complications of intraarterial monitoring include hematoma, bleeding (particularly with catheter tubing disconnections), vasospasm, arterial throm-bosis, embolization of air bubbles or thrombi, pseu-doaneurysm formation, necrosis of skin overlying the catheter, nerve damage, infection, necrosis of extremities or digits, and unintentional intraarterial drug injection. Factors associated with an increased rate of complications include prolonged cannula-tion, hyperlipidemia, repeated insertion attempts, female gender, extracorporeal circulation, the use of larger catheters in smaller vessels, and the use of vasopressors. The risks are minimized when the ratio of catheter to artery size is small, saline is continuously infused through the catheter at a rate of 2–3 mL/hr, flushing of the catheter is limited, and meticulous attention is paid to aseptic technique. Adequacy of perfusion can be continually moni-tored during radial artery cannulation by placing a pulse oximeter on an ipsilateral finger.
Clinical Considerations
Because intraarterial cannulation allows
continu-ous beat-to-beat blood pressure measurement, it is considered the
optimal blood pressure monitoring technique. The quality of the transduced
waveform, however, depends on the dynamic characteristics of
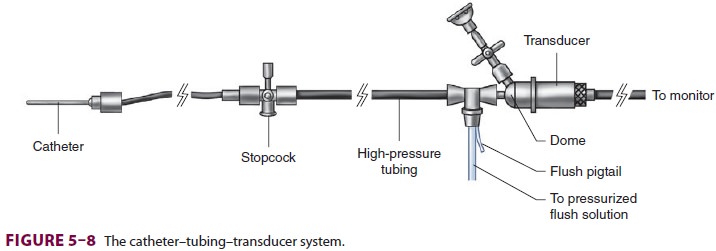
the catheter–tubing–transducer system (Figure 5–8).
False readings can lead to inappropriate therapeutic interventions.
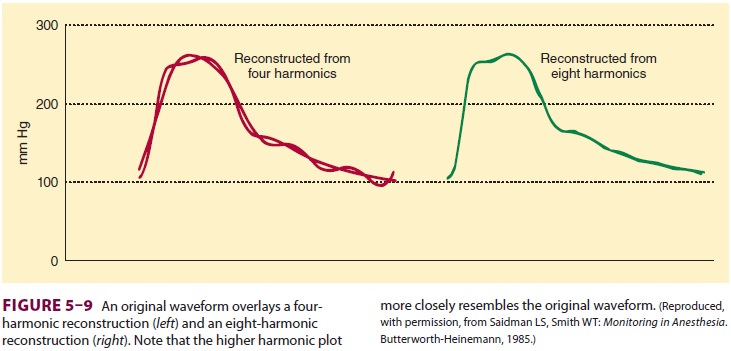
A complex waveform, such as an arterial
pulse wave, can be expressed as a summation of simple harmonic waves (according
to the Fourier theorem). For accurate measurement of pressure, the catheter–
tubing–transducer system must be capable of responding adequately to the
highest frequency of the arterial waveform (Figure 5–9). Stated another way,
the natural frequency of the measuring system must exceed the natural frequency
of the arterial pulse (approximately 16–24 Hz).
Most transducers have frequencies of
several hundred Hz (>200 Hz for disposable transducers).The
addition of tubing, stopcocks, and air in the line all decrease the frequency
of the system. If the frequency response is too low, the system will be
overdamped and will not faithfully reproduce the arterial waveform,
underestimating the sys-tolic pressure. Underdamping is also a serious problem,
leading to overshoot and a falsely high SBP.
Catheter–tubing–transducer systems must
also prevent hyperresonance, an
artifact caused by reverberation of pressure waves within the system. A damping coefficient (β) of 0.6–0.7 is
optimal. The natural frequency and damping coefficient can be determined by
examining tracing oscillations after a high-pressure flush (Figure 5–10).
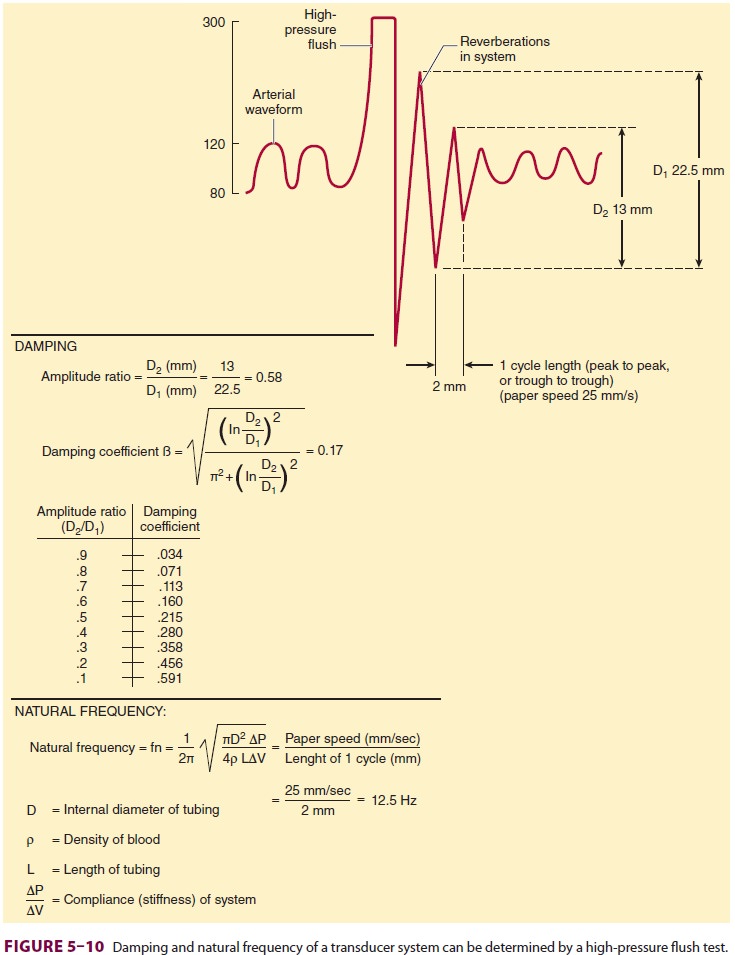
System dynamics are improved by
minimizing tubing length, eliminating unnecessary stopcocks, removing air
bubbles, and using low-compliance tubing. Although smaller diameter catheters
lower natural frequency, they improve underdampened systems and are less apt to
result in vascular compli-cations. If a large catheter totally occludes an
artery, reflected waves can distort pressure measurements.
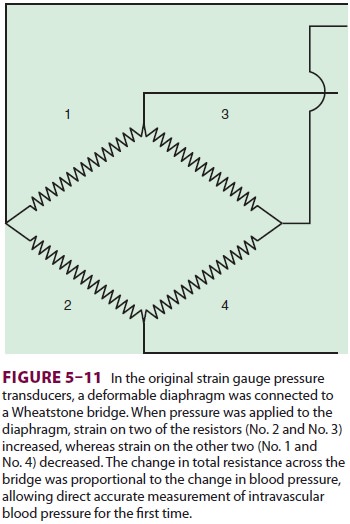
Pressure transducers have evolved from
bulky, reusable instruments to miniaturized, disposable devices. Transducers
contain a diaphragm that is distorted by an arterial pressure wave. The
mechani-cal energy of a pressure wave is converted into an electric signal.
Most transducers are resistance types that are based on the strain gauge principle: stretch-ing a
wire or silicone crystal changes its electrical resistance. The sensing
elements are arranged as a “Wheatstone bridge” circuit so that the voltage
out-put is proportionate to the pressure applied to the diaphragm (Figure 5–11).
Transducer accuracy depends on correct
cali-bration and zeroing procedures. A stopcock at the level of the desired
point of measurement—usually the midaxillary line—is opened, and the zero
trigger on the monitor is activated. If the patient’s position is altered by
raising or lowering the operating table, the transducer must either be moved in
tandem or zeroed to the new level of the midaxillary line. In a seated patient,
the arterial pressure in the brain dif-fers significantly from left ventricular
pressure. In this circumstance, cerebral pressure is determined by setting the
transducer to zero at the level of the ear, which approximates the circle of
Willis. The trans-ducer’s zero should be checked regularly, as some transducer
measurements can “drift” over time.
External calibration of a transducer
compares the transducer’s reading with a manometer, but mod-ern transducers
rarely require external calibration.
Digital readouts of systolic and
diastolic pres-sures are a running average of the highest and lowest
measurements within a certain time interval. Because motion or cautery
artifacts can result in some very misleading numbers, the arterial waveform
should always be monitored. The shape of the arterial wave provides clues to
several hemodynamic variables. The rate of upstroke indicates contractility,
the rate of downstroke indicates peripheral vascular resistance, and
exaggerated variations in size during the respiratory cycle suggest
hypovolemia. MAP is calculated by integrating the area under the pressure
curve.
Intraarterial catheters also provide
access for intermittent arterial blood gas sampling and analysis. The
development of fiberoptic sensors that can be inserted through a 20-gauge
arterial catheter enables continuous blood gas monitoring. Unfortunately, these
sensors are quite expensive and are often inaccurate, so they are rarely used.
Analysis of the arterial pressure waveform allows for estimation of cardiac
output (CO) and other hemodynamic parameters. These devices are discussed in
the section on CO monitoring.
Related Topics