Chapter: Basic & Clinical Pharmacology : Diuretic Agents
Edematous States
CLINICAL PHARMACOLOGY OF DIURETIC AGENTS
A
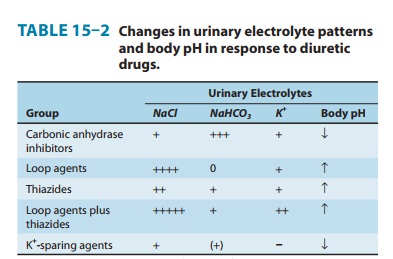
summary of the effects of diuretics on urinary electrolyte excre-tion is shown
in Table 15–2.
EDEMATOUS STATES
A
common reason for diuretic use is for reduction of peripheral or pulmonary
edema that has accumulated as a result of cardiac, renal, or vascular diseases
that reduce blood flow to the kidney. This reduction is sensed as insufficient
effective arterial blood volume and leads to salt and water retention, which
expands blood volume and eventually causes edema formation. Judicious use of
diuretics can mobilize this interstitial edema without significant reductions
in plasma volume. However, excessive diuretic therapy may compromise the
effective arterial blood volume and reduce the perfusion of vital organs.
Therefore, the use of diuretics to mobilize edema requires careful monitoring
of the patient’s hemo-dynamic status and an understanding of the
pathophysiology of the underlying illness.
HEART FAILURE
When
cardiac output is reduced by heart failure, the resultant changes in blood
pressure and blood flow to the kidney are sensed as hypovolemia and lead to
renal retention of salt and water. This physiologic response initially
increases intravascular volume and venous return to the heart and may partially
restore the cardiac output toward normal .
If
the underlying disease causes cardiac output to deteriorate despite expansion
of plasma volume, the kidney continues to retain salt and water, which then
leaks from the vasculature and becomes interstitial or pulmonary edema. At this
point, diuretic use becomes necessary to reduce the accumulation of edema,
par-ticularly in the lungs. Reduction of pulmonary vascular conges-tion with
diuretics may actually improve oxygenation and thereby improve myocardial
function. Reduction of preload can reduce the size of the heart, allowing it to
work at a more efficient fiber length. Edema associated with heart failure is
generally managed with loop diuretics. In some instances, salt and water
retention may become so severe that a combination of thiazides and loop
diuretics is necessary.
In
treating the heart failure patient with diuretics, it must always be remembered
that cardiac output in these patients is being maintained in part by high
filling pressures. Therefore, excessive use of diuretics may diminish venous
return and further impair cardiac output. This is especially critical in right
ventricular heart failure. Systemic, rather than pulmonary vascular,
conges-tion is the hallmark of this disorder. Diuretic-induced volume
contraction predictably reduces venous return and can severely compromise
cardiac output if left ventricular filling pressure is reduced below 15 mm Hg .
Reduction in cardiac output, resulting from either left or right ventricular
dysfunction, also eventually leads to renal dysfunction resulting from reduced
perfusion pressures.
Increased
delivery of salt to the TAL leads to activation of the macula densa and a
reduction in GFR by tubuloglomerular feed-back. The mechanism of this feedback
is secretion of adenosine by macula densa cells, which causes afferent arteriolar
vasoconstric-tion through activation of A1 adenosine receptors on
the afferent arteriole. This vasoconstriction reduces GFR. Tubuloglomerular
feedback–mediated reduction in GFR exacerbates the reduction that was initially
caused by decreased cardiac output. Recent work with adenosine receptor
antagonists has shown that it may soon be possible to circumvent this
complication of diuretic therapy in heart failure patients by blunting
tubuloglomerular feedback.
Diuretic-induced
metabolic alkalosis, exacerbated by hypo-kalemia, is another adverse effect
that may further compromise cardiac function. This complication can be treated
with replacement of K+ and restoration of intravascular volume with saline; however,
severe heart failure may preclude the use of saline even in patients who have
received excessive diuretic therapy. In these cases, adjunc-tive use of
acetazolamide helps to correct the alkalosis.
Another
serious toxicity of diuretic use in the cardiac patient is hypokalemia.
Hypokalemia can exacerbate underlying cardiac arrhythmias and contribute to
digitalis toxicity. This can usually be avoided by having the patient reduce Na+ intake while taking
diuretics, thus decreasing Na+ delivery to the K+-secreting collect-ing tubule. Patients who are noncompliant
with a low Na+ diet must take oral KCl supplements or a K+-sparing diuretic.
KIDNEY DISEASE AND RENAL FAILURE
A
variety of diseases interfere with the kidney’s critical role in volume
homeostasis. Although some renal disorders cause salt wasting, most cause
retention of salt and water. When renal failure is severe (GFR < 5 mL/min),
diuretic agents are of little benefit, because glomerular filtration is
insufficient to generate or sustain a natriuretic response. However, a large
number of patients, and even dialysis patients, with milder degrees of renal
insufficiency (GFR of 5–15 mL/min), can be treated with diuretics when they retain
excessive volumes of fluid between dialysis treatments.
There
is still interest in the question as to whether diuretic therapy can alter the
severity or the outcome of acute renal failure. This is because “nonoliguric”
forms of acute renal insufficiency have better outcomes than “oliguric” (<
400–500 mL/24 h urine output) acute renal failure. Almost all studies of this
question have shown that diuretic therapy helps in the short-term fluid
manage-ment of these patients with acute renal failure, but that it has no
impact on the long-term outcome.
Many
glomerular diseases, such as those associated with diabe-tes mellitus or
systemic lupus erythematosus, exhibit renal reten-tion of salt and water. The
cause of this sodium retention is not precisely known, but it probably involves
disordered regulation of the renal microcirculation and tubular function
through release of vasoconstrictors, prostaglandins, cytokines, and other
mediators. When edema or hypertension develops in these patients, diuretic
therapy can be very effective.
Certain
forms of renal disease, particularly diabetic nephropa-thy, are frequently
associated with development of hyperkalemia at a relatively early stage of
renal failure. In these cases, a thiazide or loop diuretic will enhance K+ excretion by
increasing delivery of salt to the K+-secreting collecting
tubule.
Patients
with renal diseases leading to the nephrotic syndrome often present complex
problems in volume management. These patients may exhibit fluid retention in
the form of ascites or edema but have reduced plasma volume due to reduced
plasma oncotic pressures. This is very often the case in patients with “minimal
change” nephropathy. In these patients, diuretic use may cause further
reductions in plasma volume that can impair GFR and may lead to orthostatic
hypotension. Most other causes of nephrotic syndrome are associated with
primary retention of salt and water by the kidney, leading to expanded plasma
volume and hypertension despite the low plasma oncotic pressure. In these
cases, diuretic therapy may be beneficial in controlling the volume-dependent
component of hypertension.
In
choosing a diuretic for the patient with kidney disease, there are a number of
important limitations. Acetazolamide must usually be avoided because it causes
NaHCO3 excretion and can exacerbate acidosis. Potassium-sparing
diuretics may cause hyperkalemia. Thiazide diuretics were previously thought to
be ineffective when GFR falls below 30 mL/min. More recently, it has been found
that thiazides, which are of little ben-efit when used alone, can be used to
significantly reduce the dose of loop diuretics needed to promote diuresis in a
patient with GFR of 5–15 mL/min. Thus, high-dose loop diuretics (up to 500 mg
of furosemide/d) or a combination of metolazone (5–10 mg/d) and much smaller
doses of furosemide (40–80 mg/d) may be useful in treating volume overload in
dialysis or predialysis patients. There has been some interest in the use of
osmotic diuretics such as mannitol, because this drug can shrink swollen
epithelial cells and may theoretically reduce tubular obstruction.
Unfortunately, there is no evidence that mannitol can prevent ischemic or toxic
acute renal failure. Mannitol may be useful in the management of hemoglobinuria
or myoglobinuria. Lastly, although excessive use of diuretics can impair renal
function in all patients, the consequences are obviously more serious in
patients with underlying renal disease.
HEPATIC CIRRHOSIS
Liver
disease is often associated with edema and ascites in con-junction with
elevated portal hydrostatic pressures and reduced plasma oncotic pressures.
Mechanisms for retention of Na+ by the kidney in this setting include diminished renal
perfusion (from systemic vascular alterations), diminished plasma volume (due
to ascites formation), and diminished oncotic pressure (hypoalbu-minemia). In
addition, there may be primary Na+ retention due to elevated plasma aldosterone levels.
When
ascites and edema become severe, diuretic therapy can be very useful. However,
cirrhotic patients are often resistant to loop diuretics because of decreased
secretion of the drug into the tubular fluid and because of high aldosterone
levels. In contrast, cirrhotic edema is unusually responsive to spironolactone
and eplerenone. The combination of loop diuretics and an aldosterone receptor
antagonist may be useful in some patients. However, considerable caution is
necessary in the use of aldosterone antago-nists in cirrhotic patients with
even mild renal insufficiency because of the potential for causing serious
hyperkalemia.
It
is important to note that, even more than in heart failure, overly aggressive
use of diuretics in this setting can be disastrous. Vigorous diuretic therapy
can cause marked depletion of intra-vascular volume, hypokalemia, and metabolic
alkalosis. Hepatorenal syndrome and hepatic encephalopathy are the unfortunate
consequences of excessive diuretic use in the cir-rhotic patient.
IDIOPATHIC EDEMA
Idiopathic
edema (fluctuating salt retention and edema) is a syn-drome found most often in
20–30 year-old women. Despite intensive study, the pathophysiology remains
obscure. Some stud-ies suggest that surreptitious, intermittent diuretic use
may actu-ally contribute to the syndrome and should be ruled out before
additional therapy is pursued. While spironolactone has been used for
idiopathic edema, it should probably be managed with moder-ate salt restriction
alone if possible. Compression stockings have also been used but appear to be
of variable benefit.
Related Topics