Chapter: 11th Geography : Chapter 6 : Atmosphere
Composition of the Atmosphere
Composition
of the Atmosphere
The atmosphere is essential for the survival of all
the organisms on the earth. The atmosphere is a blanket of gases and suspended
particles that entirely envelope the earth. It extends outward over thousands
of kilometres from the earth’s
Water vapour, aerosols and tiny solid
particles occur in varying quantities as suspended material. These are
responsible for weather phenomena as they have ability to absorb and release
heat energy.
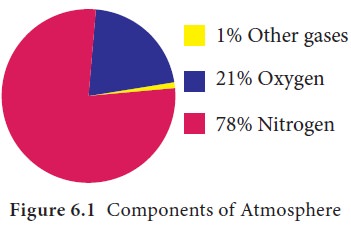
The atmosphere is composed of mixture of many gases,
water vapour and other solid particles. The major components are nitrogen
(78%), oxygen (21%) and other gases (1%). Argon, Carbon dioxide, Neon and the
other gases found in the atmosphere (Figure 6.1).
Layers of the Atmosphere
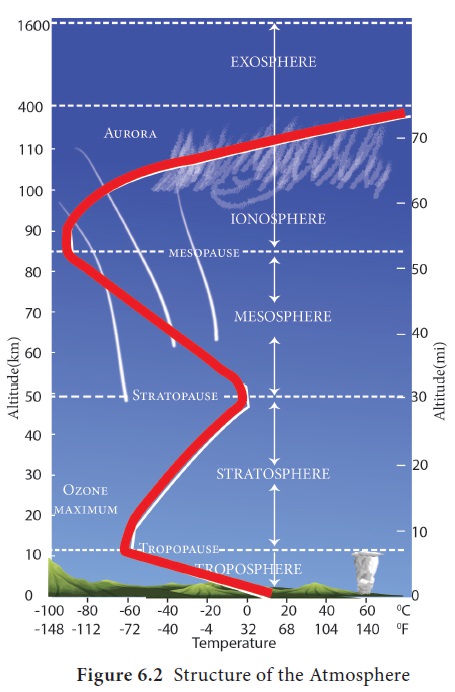
The atmosphere is divided into five distinct layers
(Figure.6.2) based on the temperature variations. They are,
1.
Troposphere
2.
Stratosphere
3.
Mesosphere
4.
Ionosphere(
Thermosphere) and
5.
Exosphere
Troposphere
The troposphere( Figure 6.2) is the lower most
layer of the atmosphere. It extends approximately to a height of 8 km from the
poles and 18 km from the equator. The height of the troposphere changes
seasonally also. It increases during summer and decreases during winter.
All weather phenomena occur in this layer as it has
dust particles and water vapour. This layer has clouds which produce
precipitation on the earth. The Sun’s rays directly fall on the earth and then
they are reflected back into the atmosphere. The temperature decreases in the
troposphere with increase in altitude at the rate of 1 ° C for 165 metre or 6.5 ° C for every 1000 metres of ascent. This is
known as lapse rate of temperature.
This is the densest layer as it contains 70 to 80 percent of gases. The outer
boundary of the troposphere is called tropopause, which is about 1.5 kilmeter
thick.
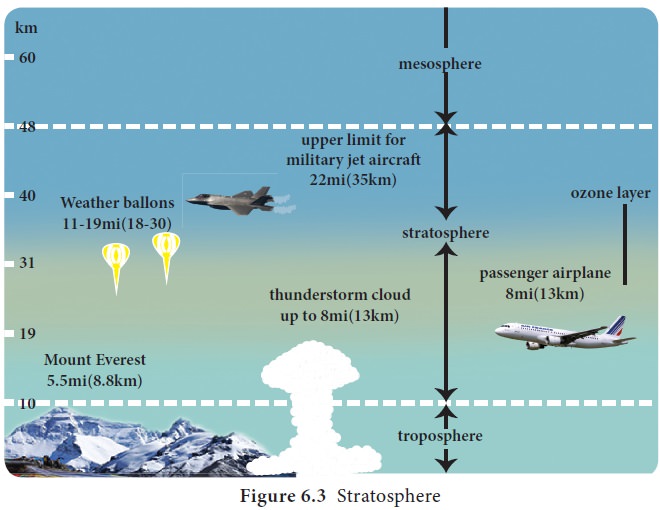
Stratosphere
It is the second layer of the atmosphere found
above the troposphere. It approximately extends up to a height of 50 km from
the earth’s surface. Temperature is constant up to a height of 20 km and
increases gradually up to the stratopause where temperature is nearly -4° C.
The lower part of this layer is highly concentrated with ozone gas which is
called as ‘ozonosphere’. It prevents
the ultra-violet rays from the Sun to enter into the lower part of the
atmosphere as the rays are highly harmful it causes skin cancer and other ill
effects to living organisms. But the ozone layer safeguards the life on the
earth.
Mesosphere
The mesosphere is the third layer of the atmosphere
found approximately up to a height of 85 km above the surface of the earth. It
is the coldest layer of the atmosphere. The temperature decreases with increase
of altitude due to the absence of ozone. Its upper boundary is called mesopause where temperature reaches
2908C. Luminous noctilucent clouds form here due to the presence of cosmic
dust. Meteors falling from the space get burned in this layer. It is because
when meteors hit the air, the air gets compressed and heated up causing meteors
to burn out.
Ionosphere (Thermosphere)
The ionosphere is the fourth layer of the
atmosphere extending approximately up to a height of 400 km. The temperature
increases rapidly up to 1,0008C. It is due to the absorption of very short wave
and high energy solar radiation by the atoms of hydrogen and oxygen gases. When
light energy is transformed into heat energy, some gas molecules lose or gain
electrons and become the charged particles called ions. The charged particles
forming the lower part of the thermosphere as a zone, is called Ionosphere
(Figure 6.4). These ionised particles create auroras at higher latitudes.
Ionosphere can reflect radio waves back to the earth. This facilitates long
distance wireless satellite communication. The credit of discovering ionosphere
goes to Hennelly and Heaviside.
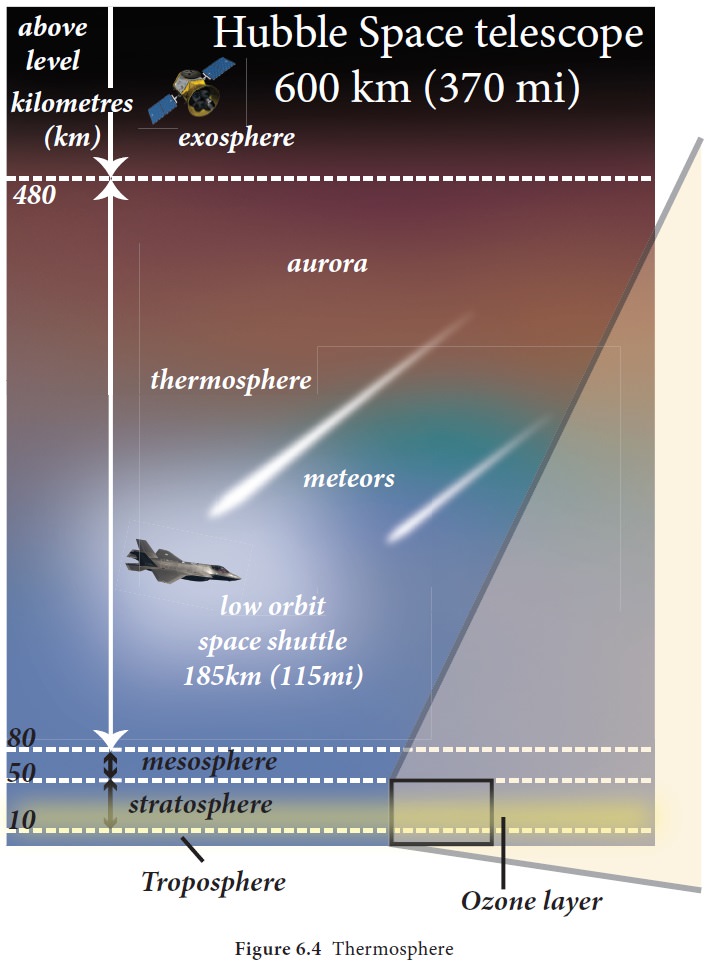
Exosphere
The upper most layer of the atmosphere which
extends into the outer space from above 400 km up to 1600km. It has rarefied
contents. It contains mainly oxygen and hydrogen atoms. These atoms can travel
hundreds of kilometres without colliding with one another. Thus, the exosphere
has no longer behaves like a gas. The temperature increases with increase of
altitude and it ranges as high as 1650 ° C. The gravitational pull is minimal
in this layer. This layer gradually merges with the space.
Ozone and Ozone Depletion
Ozone (O3) is form of oxygen that combines three atoms into each molecule. It absorbs and filters the harmful ultraviolet B radiation coming from the sun. This way the ozone layer protects all life on earth. However, ozone is harmful when it develops near the ground. It causes health problems like asthma and other respiratory illness.
Ozone
Depletion: A steady decline in the
concentration of ozone in the
earth’s stratosphere (the ozone layer)
is called ozone depletion.
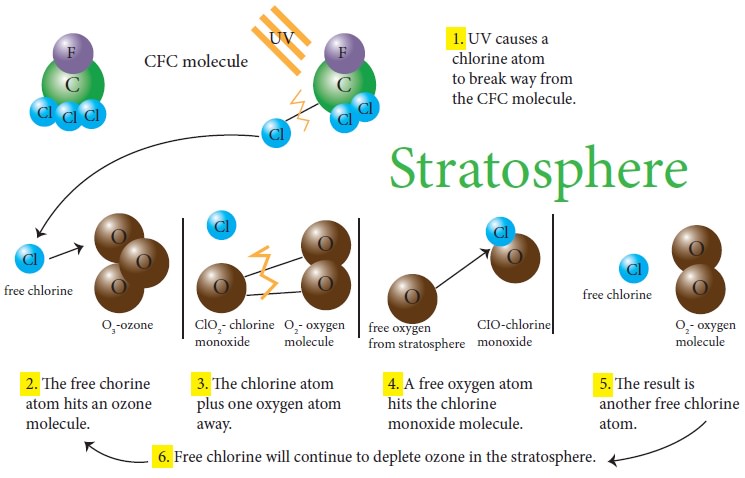
Ozone depletion
occurs when chloro fluoro
carbon (CFC) and halon gases, formerly
found in aerosol
spray cans and refrigerants
are released into
the atmosphere and they
cause chemical reactions that break down ozone molecules and reduce the
concentration of them. Nitrogen oxide released by emitted by supersonic
aircrafts can also destroy the ozone molecules to break down. Ozone-depleting
substances are present throughout the stratospheric ozone layer because they
are transported great distances by atmospheric air motions. The severe
depletion of the Antarctic ozone layer known as the “ozone hole” occurs because
of the special atmospheric and chemical conditions that exist there and nowhere
else on the globe. The very low winter temperatures in the Antarctic
stratosphere cause polar stratospheric clouds (PSCs) to form. Special reactions
that occur on PSCs, combined with the relative isolation of polar stratospheric
air, allow chlorine and bromine reactions
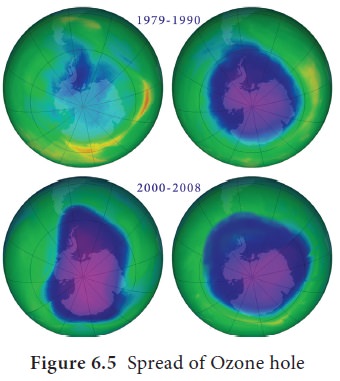
Satellite images of the earth over
last decades observed that the atmospheric ozone layer is getting thinner. On
October 2, 2015, the ozone hole was recorded to its maximum size of 28.2
million sq.km over Antarctica
(Figure 6.5). The size of the ozone hole is larger than the size of continent
of North America. The ozone holes over Antarctica allow the ultraviolet
radiation to enter and cause global warming, skin cancer, eye cataract and even
blindness.
Depletion
of the ozone layer
has consequences on human, animal, plants
and micro organisms. This typically results from higher UV levels reaching us
on earth. Research confirms that high levels of UV rays cause non-melanoma skin
cancer.
To protect the ozone layer for our
future generation, avoid using products which are emitting pollutants such as
aerosol sprays, blowing agents for foams and packing materials, as solvents and
as refrigerants.
Related Topics